Estimate the LOC of ethylene using Equations 6-15 and 6-16 in the textbook. Compare to the experimental
Question:
Estimate the LOC of ethylene using Equations 6-15 and 6-16 in the textbook. Compare to the experimental value in Table 6-3.
Table 6-3:
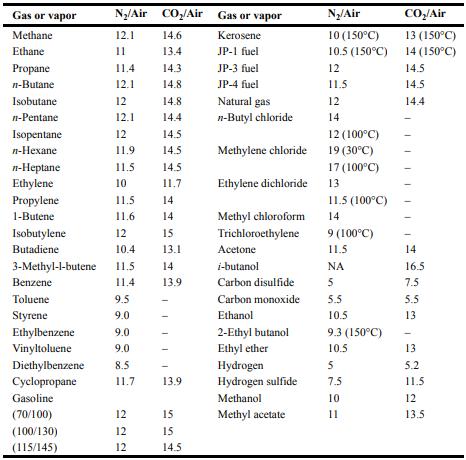
Equation 6-15:
![]()
Equation 6-16:
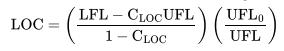
Transcribed Image Text:
Gas or vapor Methane Ethane Propane n-Butane Isobutane n-Pentane Isopentane n-Hexane 12 11.9 11.5 10 11.5 11.6 12 10.4 3-Methyl-l-butene 11.5 11.4 n-Heptane Ethylene Propylene 1-Butene Isobutylene Butadiene Benzene Toluene Styrene Ethylbenzene Vinyltoluene N/Air CO/Air 12.1 14.6 11 13.4 11.4 14.3 12.1 14.8 12 14.8 12.1 14.4 14.5 14.5 Gasoline (70/100) (100/130) (115/145) 9.5 9.0 9.0 9.0 Diethylbenzene 8.5 Cyclopropane 11.7 12 222 12 12 14.5 11.7 14 14 15 13.1 14 13.9 1 13.9 15 15 14.5 Gas or vapor Kerosene JP-1 fuel JP-3 fuel JP-4 fuel Natural gas n-Butyl chloride Methylene chloride Ethylene dichloride Methyl chloroform Trichloroethylene Acetone i-butanol Carbon disulfide Carbon monoxide Ethanol 2-Ethyl butanol Ethyl ether Hydrogen Hydrogen sulfide Methanol Methyl acetate N/Air 10 (150C) 10.5 (150C) 12 11.5 12 14 12 (100C) 19 (30C) 17 (100C) 13 11.5 (100C) 14 9 (100C) 11.5 5 5.5 10.5 9.3 (150C) 10.5 5 7.5 10 11 COy/Air 13 (150C) 14 (150C) 14.5 14.5 14.4 1 14 16.5 7.5 5.5 13 13 5.2 11.5 12 13.5
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Equations 615 and 616 can be used to estimate the Limiting Oxygen Concentration LOC of ethylene by u...View the full answer

Answered By

Mercy Kangai
I provide creative and detailed administrative, web search, academic writing, data entry, Personal assistant, Content writing, Translation, Academic writing, editing and proofreading services. I excel at working under tight deadlines with strict expectations. I possess the self-discipline and time management skills necessary to have served as an academic writer for the past seven years. I can bring value to your business and help solve your administrative assistant issues. I have extensive experience in marketing and small business management.
4.80+
27+ Reviews
82+ Question Solved
Related Book For 

Chemical Process Safety Fundamentals With Applications
ISBN: 9780134857770
4th Edition
Authors: Daniel A. Crowl, Joseph F. Louvar
Question Posted:
Students also viewed these Engineering questions
-
Estimate the LOC for (a) carbon monoxide and (b) heptane using Equation 6-15. Compare to experimental values in Table 6-3. Table 6-3: Equation 6-15: Gas or vapor Methane Ethane Propane n-Butane...
-
In a study of hypnotic suggestion, 16 male volunteers were randomly allocated to an experimental group and a control group. Each subject participated in a two-phase experimental session. In the first...
-
To calculate variance and standard deviation, we take the deviations from the mean. At times, we need to consider the deviations from a target value rather than the mean. Consider the case of a...
-
The following scatter plot indicates that 200 150 > 100 50 0 0 20 X 40 O a log x transform may be useful Oa y transform may be useful a x transform may be useful Ono transform is needed Oa 1/x...
-
Determine the volume and the surface area of the chain link shown knowing that it is made from a 0.5-in.-diameter bar and that R = 0.75in and L = 3in.
-
Given the following information, how many units must be sold to achieve a profit of $25,000? Direct labor hours:..................................0.2 hour/unit Direct labor...
-
Gilead is a large drug producer, with a majority of its prescription drug product sales occurring in the United States. Gilead produces anti-HIV drug therapies, including the drugs Atripla, Truvada,...
-
Using the following per-unit and total amounts, prepare a flexible budget at the 14,000-, 15,000-, and 16,000-unit levels of production and sales for Celestial Products, Inc.: Selling price per unit...
-
Why did big business get bigger in the US than in other industrialized countries (e.g., Britain and Germany) during the 2d Industrial Revolution? How did the US differ from other countries such as...
-
Draw an approximate flammability triangle diagram for methyl alcohol. Use published flammability data from Appendix B and Table 6-3. If a gas containing 20\% methyl alcohol, \(5 \%\) oxygen, and \(75...
-
Estimate the UFL and the LFL for ethylene using the stoichiometric concentrations and Equations 6-10 and 6-11 in the textbook. Compare to the experimental values in Appendix B. Data From Appendix B:...
-
An investor considers investing $10,000 in the stock market. He believes that the probability is 0.30 that the economy will improve, 0.40 that it will stay the same, and 0.30 that it will...
-
In your state, whose responsibility is it to view and confirm a vendor/sellers identity?
-
A wind turbine is rotating of diameter 90 m is rotating at under steady winds flowing through the turbine at a rate of 42,000 kg/s. if 180 kW power is produced by the turbine, determine the: a)...
-
List two (2) techniques and strategies for tracking legislative amendments.
-
Write a report about what happened at The January 6th hearings. The Report must have: A tight focus. Accurate information. Synthesis of information. Clear definitions of terms. NO personal opinion....
-
Knowing Apple's current market position, Look out over the next three to five years. What is your overall assessment of the following? Volatility of the focal company's supply chain risk profile;...
-
At the end of the current year, Accounts Receivable has a balance of $2,500,000; Allowance for Doubtful Accounts has a debit balance of $9,000; and net sales for the year total $32,000,000. Using the...
-
Walker, Inc., is an all-equity firm. The cost of the company's equity is currently 11.4 percent and the risk-free.rate is 3.3 percent. The company is currently considering a project that will cost...
-
The amount of power transmitted by sunlight depends on latitude and the surface area of the solar collector. On a clear day at a certain northern latitude, 0.6 kW/m 2 of solar power strikes the...
-
The property of a fluid called viscosity is related to its internal friction and resistance to being deformed. The viscosity of water, for instance, is less than that of molasses and honey, just as...
-
Referring to the description in Problem P3.16, and given that the viscosity of a certain engine oil is 0.25 kg/(m s), determine the value in the units (a) Poise and (b) Slug/(ft s). Problem P3.16...
-
A scraper whose weight on the driving wheels is 16,330 kg has a gross weight 29,030 kg. If the road surface is wet earth having a rolling resistance factor of 60 kg/t, what is the maximum grade the...
-
What distinguishes health care economics from that of other sectors? Explain.
-
Flight Caf prepares in-flight meals for airlines and its planning budget for July appears below: Flight Caf Planning Budget For the Month Ended July 31 Budgeted meals (q) Revenue ($4.10q) Expenses:...

Study smarter with the SolutionInn App


