An aqueous feed of A and B (400 liter/min, 100 mmol A/liter, 200 mmol B/liter) is to
Question:
An aqueous feed of A and B (400 liter/min, 100 mmol A/liter, 200 mmol B/liter) is to be converted to product in a mixed flow reactor. The kineticsof the reaction are represented by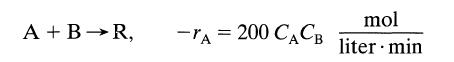
Find the volume of reactor needed for 99.9% conversion of A to product.
Transcribed Image Text:
A + B →R, -A = 200 CACB mol liter min
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (3 reviews)
Calculate the feed rate of A FA 400 litermin 100 mmolliter 40000 mmol...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
An aqueous feed of A and B (400 liter/min, 100 mmol A/liter, 200 mmol B/liter) is to be converted to product in a plug flow reactor. The kinetics of the reaction is represented by Find the volume of...
-
Art-labeling Activity: Internal anatomy of the heart (1 of 2) Part A Drag the labels to identify structural components of the heart. Left ventricle Inferior vena cava Pulmonary trunk Right ventricle...
-
There is a crop that the value p next month is random. The value can be either small p=1 or large P=3 depending on the weather. Half of people are optimists believing the value of the crop will be...
-
If more customers want to borrow funds at the prevailing interest rate, a financial institution can increase its profits by raising interest rates on its loans. Is this statement true, false, or...
-
Ortiz Company produced 10,000 units during the past year but sold only 8,500 of the units. The following additional information is also available: Direct materials used...
-
The table at the left shows the sample ages of students in a class. Find the mean, median, and mode of the ages. Are there any outliers? Which measure of central tendency best describes a typical...
-
In Fig. 29.23 the capacitor plates have area 5.00 cm2 and separation 2.00 mm. The plates are in vacuum. The charging current ic has a constant value of 1.80mA. At t = 0 the charge on the plates is...
-
A car runs on wheels of diameter 3 5 0 mm . The car is travelling at an average speed of 7 5 km / h , what is the angular velocity of the wheels?
-
Enzyme E catalyses the fermentation of substrate A (the reactant) to product R. Find the size of mixed flow reactor needed for 95% conversion of reactant in a feed stream (25 liter/min) of reactant...
-
A gaseous feed of pure A (2 mol/liter, 100 mol/min) decomposes to give a variety of products in a plug flow reactor. The kinetics of the conversion is represented by Find the expected conversion in a...
-
Graph the three-month Treasury bill interest rate, the ten-year government bond interest rate, and the CPI inflation rate (based on the percentage change in the CPI from one year earlier) on the same...
-
3. Is Apple's decision to reduce packaging for the iPhone 12 a good marketing decision? (Is it responsible? Will it strengthen relationships with customers? Will it provide other benefits to Apple or...
-
Moody Corporation uses a job - order costing system with a plantwide predetermined overhead rate based on machine - hours. At the beginning of the year, the company made the following estimates:...
-
write the steps to enable Remote Direct Memory Access (RDMA) with Switch Embedded Teaming (SET).
-
(a) (b) Give ONE example for EACH of these categories of vehicle options mechanical device and convenience option. (4 marks) Isabella owns a 2022 Toyota Camry and its gasoline consumption is 7.9...
-
Briefly explain each of the following terms and give examples of how they can be applied (or how they can occur) in a work environment Risk Management Risk Governance External Audit The Control...
-
What rule should the firm use in deciding when to shut down production in the short run? In the long run?
-
The liquidliquid extractor in Figure 8.1 operates at 100F and a nominal pressure of 15 psia. For the feed and solvent flows shown, determine the number of equilibrium stages to extract 99.5% of the...
-
Also Hall of Fame Problem. The irreversible liquid-phase reactions Reaction(1)A + B 2Cr1C = k1CCACBReaction(2)2B+CDr2D=k2DCBCC are carried out in a PFR with heat exchange. The temperature profiles...
-
The reaction A+B2C is carried out in a packed-bed reactor. Match the following temperature and conversion profiles for the four different heat-exchange cases: adiabatic, constant Ta, cocurrent...
-
A reaction is to be carried out in the packed-bed reactor shown in Figure P12-19C. PFR with heat exchange. The reactants enter the annular space between an outer insulated tube and an inner tube...
-
Suppose you own 5,000 shares of Abbvie (Ticker: ABBV) in your portfolio and want to sell some shares. The current price for ABBV is $78 per share. Your valuation model indicates that this share price...
-
1) Investment X yields a payment of $8000 at the end of year 1, a payment of $8000 at the end of year 2, a payment of $8000 at the end of year 3, and a payment of $12000 at the end of year 4....
-
Assuming an investor bought at the current market price, what is the expected rate of return to SRT bonds? Is the expected rate of return different from the YTM? Why or why not? Maturity is 1 year,...
Probability An Introduction With Statistical Applications 2nd Edition - ISBN: 1118947088 - Free Book

Study smarter with the SolutionInn App


