Question: Find the conversion after 1 hour in a batch reactor for A R, -A= 3C05 mol liter.hr' CA0 = 1 mol/liter
Find the conversion after 1 hour in a batch reactor for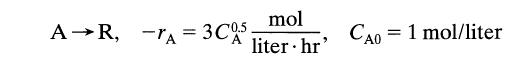
A R, -A= 3C05 mol liter.hr' CA0 = 1 mol/liter
Step by Step Solution
★★★★★
3.36 Rating (162 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

To find the conversion after 1 hour in a batch reactor for the given reaction we need to integrate the rate law to find the relation between concentration and time The provided image gives the reactio... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock


