Find the overall order of the irreversible reaction from the following constant-volume data using equimolar amounts of
Question:
Find the overall order of the irreversible reaction
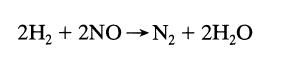 from the following constant-volume data using equimolar amounts of hydrogen and nitric oxide:
from the following constant-volume data using equimolar amounts of hydrogen and nitric oxide:
Transcribed Image Text:
2H₂ + 2NO N₂ + 2H₂O
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
To find the overall order of the reaction we can use the following equation rate kHnNOm where k is t...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
The following data where measured for the reaction BF3(g) + NH3(g) -- F3BNH3(g): (a) What is the rate law for the reaction? (b)What is the overall order of the reaction? (c) Calculate the rate...
-
1. Hydrogen peroxide reacts with thiosulfate ion in slightlyacidic solution as follows: H 2 O 2 + 2S 2 O 3 2? + 2 H + ? 2H 2 O + S 4 O 6 2? The reaction rate is independent of the hydrogen...
-
Can you say GDP is underestimated or overestimated using the Expenditures approach to calculating GDP? Why?
-
What does it mean that flame is non-luminous it is yellow it is invisible It has a very bright blue inner cone it is dark
-
A hedger takes a short position in five T-bill futures contracts at the price of 98 5/32. Each contract is for $100,000 principal. When the position is closed, the price is 95 12/32. What is the gain...
-
Amherst City provides a defined benefit pension plan for employees of the city electric utility, an enterprise fund. Assume that the projected level of earnings on plan investments is $190,000, the...
-
A proximity probe has a useful frequency range related to what operating parameter?
-
1. Describe the marketing exchange relationship that exists between Southwest Airlines and its customers. 2. Describe the 5Cs of the marketing framework as they pertain to Southwest Airlines. 3. How...
-
Problem #4: Air at 100 kPa and 300 K flows steadily through a square duct with side length of 0.5 m. The air enters with a uniform speed of 5 m/s. An electric surface heating element is attached to...
-
Aqueous A reacts to form R (A R) and in the first minute in a batch reactor its concentration drops from C A0 = 2.03 mol/liter to C Af = 1.97 mol/liter. Find the rate equation for the reaction if...
-
Snake-Eyes Magoo is a man of habit. For instance, his Friday evenings are all alike-into the joint with his week's salary of $180, steady gambling at "2-up" for two hours, then home to his family...
-
Is the mean cost of food items in a "typical" basket of products the same across Canada? The accompanying list shows the costs of the same items when purchased in four different cities. Do these...
-
A dc battery can deliver 45 mA at 9 V. What is the power rating?
-
Tanya and Luca are married. They are not self-employed and work in New Jersey. They take a standard deduction on their taxes. Tanya earns $156,750/year; Luca earns $227,210/year. They have the...
-
If an apartment complex will need painting in 3 years and the job will cost $115,000, what amount needs to be deposited into an account now in order to have the necessary funds? The account pays 2%...
-
Joshua purchases 100 shares of McDonalds (MCD) stock on April 13, 2015 at $83.98 per share. Three years later on April 13, Joshua sells the 100 shares for $152.12 per share. In addition, Joshua...
-
What are some methods that you use to ensure accurate information is being inputted into devices and systems that you use?
-
Inevitably, a firm in monopolistic competition ends up producing where its ATC curve is tangent to its demand curve. Explain.
-
A Bloomberg Businessweek subscriber study asked, In the past 12 months, when traveling for business, what type of airline ticket did you purchase most often? A second question asked if the type of...
-
Describe how would your reactor volume and number of reactors change if you only needed 50% conversion to produce the 200 million pounds per year required?
-
If it takes 11 minutes to cook spaghetti in Ann Arbor, Michigan, and 14 minutes in Boulder, Colorado, how long would it take in Cuzco, Peru? Discuss ways to make the spaghetti more tasty. If you...
-
How do the steps in the design of a CSTR differ from those of a CSTR or a PFR with pressure drop?
-
Negotiation Report #1 Paint Manufacturer Negotiation Report Paint Manufacturer 1. Did you reach a settlement of the dispute? If so, state the terms of that settlement. 2. What was your Batna? 3. 4....
-
Discuss the possible characteristics and profile of a person who commits occupational fraud. Then further discuss how behavioral red flag indicators of fraud can be used to detect and prevent...
-
separate sheets provided. No additional sheets are required and they won't be gi 1. a. A primitive transport protocol uses only two sequence numbers and implements lossless order- preserving delivery...

Study smarter with the SolutionInn App


