Based on these molecular views, determine whether each pictured base is weak or strong. NH3- NH4+ (a)
Question:
Based on these molecular views, determine whether each pictured base is weak or strong.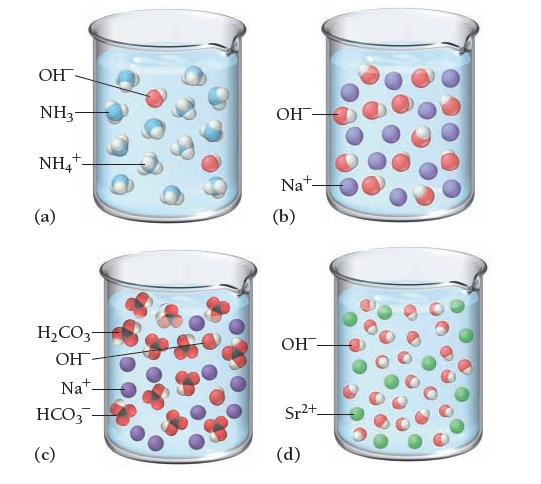
Transcribed Image Text:
ОН NH3- NH4+ (a) H2CO3- ОН- Nat- HCO3- (c) OH Nat- (b) OH- Sr2+_ (d)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a Beaker A shows that ammonia NH3 and ammonium hydroxide NH4OH both are ...View the full answer

Answered By

Benish Ahmad
I'm a professional software engineer. I'm lectutrer at GCUF and I have 3 years of teaching experience. I'm looking forward to getting mostly computer science work including:
Programming fundamentals
Object oriented programming
Data structures
object oriented design and analysis
Database system
Computer networks
Discrete mathematics
Web application
I am expert in different computer languages such as C++, java, JavaScript, Sql, CSS, Python and C#. I'm also have excellent knowledge of essay writing and research. I have worked in other Freelancing website such as Fiverr and Upwork. Now I have finally decided to join the SolutionInn platform to continue with my explicit work of helping dear clients and students to achieve their academic dreams. I deliver plagiarism free work and exceptional projects on time. I am capable of working under high pressure.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Based on these molecular views, determine whether each pictured acid is weak or strong. H3O+- HF- F (a) CHO H3O+- HCHO2- (c) HO+- (b) I- NO3- HO+ (d)
-
Read the case study "Southwest Airlines," found in Part 2 of your textbook. Review the "Guide to Case Analysis" found on pp. CA1 - CA11 of your textbook. (This guide follows the last case in the...
-
Emerson Electric Company was founded in 1890 as a manufacturer of motors and fans. In 1993, Emerson marked its thirty-sixth consecutive year of improved earnings per share. On $8.2 billion sales, the...
-
How are the interface and implementation sections of an Objective-C class specified?
-
Explain how the two-, three-, and four-variance overhead analyses are related.
-
At the end of January 2010, the records of Atlanta Company showed the following for a particular item that sold at $17 per unit: Required: 1. Assuming the use of a periodic inventory system, prepare...
-
Under what circumstances does a nonparty have an absolute right to intervene?
-
Reservoir Bottling Company reported the following information at the end of the year. Total current assets are worth $237,513 at book value and $219,344 at market value. In addition, plant and...
-
3) Cass SA, a leading wind energy developer, is planning an acquisition of City BV, a cable company. Cass is offering City's shareholders 0.25 and 0.8 shares of Cass per share of City. Current...
-
The binding of oxygen by hemoglobin in the blood involves the equilibrium reaction: In this equation, Hb is hemoglobin. The pH of normal human blood is highly controlled within a range of 7.35 to...
-
Identify the Lewis acid and Lewis base from among the reactants in each equation. a. Ag (aq) + 2NH3(aq) Ag(NH3)2+(aq) b. AlBr3 + NH3 H3NAIBr3 C. F (aq) + BF3(aq) = BF4 (aq)
-
NCR Corporation discloses the following in notes to its 2007 10-K report. a. Describe the difference between a spin-off and a split-off. b. What effects did NCR's spin-off of Teradata have on NCR's...
-
Business Analytics Inventory Model Barilla Pasta Case Background (This case was taken from an article that appears in The Economist Nov. 2020) Barilla Pasta is the largest pasta maker in the world....
-
171. In hardy-weinberg equation, the frequency of all homozygous individuals is represented by. (1) p (3) 2pq (2) q (4) p+q 172. Excess use of herbicides, Pesticides has only resulted in selection of...
-
Hiring Ethics: A Case of Undue Influence in Employee Selection Case Author: Salvador G. Villegas & Kristi M. Bockorny Online Pub Date: January 03, 2022 | Original Pub. Date: 2020 Subject: Business...
-
Find the missing endpoint if S is the midpoint RT. R(-9, 4) and S(2, -1); Find 7. S(-4, -6) and T(-7, -3); Find R. B is the midpoint of AC and E is the midpoint of BD. If A(-9, -4), C(-1, 6), and...
-
Cost behavior Merry Olde Games produces croquet sets. The company makes fixed monthly payments to the local utility based on the previous year's electrical usage. Any difference between actual and...
-
A firm has $450,000 in funds to distribute to its unsecured creditors. Three possible sets of unsecured creditor claims are presented. Calculate the settlement, if any, to be received by each...
-
Determine by direct integration the values of x for the two volumes obtained by passing a vertical cutting plane through the given shape of Fig. 5.21. The cutting plane is parallel to the base of the...
-
In recent years, a number of nearby stars have been found to possess planets. Suppose the orbital radius of such a planet is found to be 4.0 10 11 m, with a period of 1100 days. Find the mass of the...
-
A newly discovered asteroid is found to have a circular orbit, with a radius equal to 27 times the radius of the Earths orbit. How long does this asteroid take to complete one orbit around the Sun?
-
The region of the solar system between Mars and Jupiter contains many asteroids that orbit the Sun. Consider an asteroid in a circular orbit of radius 5.0 10 11 m. Find the period of the orbit.
-
Assume an organic compound has a partition coefficient between water and ethyl acetate equal to 8.12. If there are initially 7.10 grams of the compound dissolved in 75.0 mL of water, how many grams...
-
NO. DATE Use the table below Write a Java program that will display the output below. code may vary... For your reference QUANTITY ITEM PRICE 1 Bond Paper (rim) 230 1 Notebook 30 1 1 Printer Ink 5500...
-
Build a GUI conference registration program: StuPre | Conference Registration Event # of Participants Select Event 1 1 Event 2 2 Event 3 0 Event 4 1 Event 5 0 Enter your email: Register

Study smarter with the SolutionInn App


