Calculate the free energy change for this reaction at 25 C. Is the reaction spontaneous? C3H8(g) +
Question:
Calculate the free energy change for this reaction at 25 °C. Is the reaction spontaneous?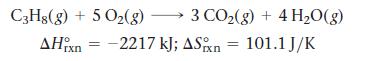
Transcribed Image Text:
C3H8(g) + 5O₂(g) 5 O₂(g) → 3 CO₂(g) + 4H₂O(g) AHin = -2217 kJ; ASixn 101.1 J/K rxn
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
2247...View the full answer

Answered By

Pranav Makode
I am a bachelor students studying at professor ram meghe institute of technology and research. I have a great experience of being an expert. I have worked as an expert at helloexperts and solvelancer as a part time job. I have also worked as a doubt solver at ICAD SCHOOL OF LEARNING, which is in Amravati city. I have also worked as an Freelancer.
I have great experience of helping students, as described above. I can help any students in a most simple and understandable way. I will not give you have any chance for complaint. You will be greatfull to accept me as an expert.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Calculate the free energy change for this reaction at 25 C. Is the reaction spontaneous? 2 Ca(s) + O2(g) 2 CaO(s) 1269.8 kJ; .Sixn -364.6J/K , xn
-
Calcium oxide, CaO, is manufactured by decomposition of calcium carbonate, CaCO3, in a furnace at 500 K: CaCO3(s)?CaO(s)+CO2(g) The spontaneity of a reaction can be determined from the free energy...
-
You monitor the reaction of a protein forming a dimer and find an apparent rate constants of 6 s-1 with 1 M total protein and 10 s-1 with 5 M total protein. a. What are the forward and reverse rate...
-
How can staff review the effectiveness of their work, the services they provide and the social and cultural factors impacting on clients, groups or communities?
-
Discuss the significance of economic development to international marketing. Why is the knowledge of economic development of importance in assessing the world marketing environment? Discuss.
-
Trans American Airlines is a large domestic airline servicing all major cities throughout the United States. It uses control systems for many of its activities, including airplane maintenance,...
-
Hooters Restaurant in Myrtle Beach, South Carolina, used an alternative dispute resolution program, a program to resolve disputes outside the traditional court system. Employees of Hooters had to...
-
Selected T-accounts of Moore Company are given below for the just completed year: Required: 1. What was the cost of raw materials put into production during the year? 2. How much of the materials in...
-
Jobs R Us, Inc. Is a recruiting firm that specializes in post- college placement in the finance industry. Its clients are currently concentrated in the North-Eastern United States lt is contemplating...
-
Fill in the blanks in the table. Both H and S refer to the system. T AS + AG Temperature dependent Low Temperature Spontaneous High Temperature Spontaneous Nonspontaneous Nonspontaneous
-
Calculate the change in Gibbs free energy for each of the sets of H rx n , S rx n , and T given in Problem 42. Predict whether or not each reaction is spontaneous at the temperature indicated....
-
How would you use training for spouses of expatriate employees to improve employee performance and the duration of service overseas?
-
HUMAN RELATION. For this assignment, you will need to monitor your emotions over the course of a day. Be sure to check your emotions at least 12 times during that day. Make a chart of your emotions...
-
Discuss FOUR (4) principles that have a strong, sustainable and positive reputation of brand. Provide ONE (1) relevant example for each principle. You must include the references.
-
How does Data analytics impact the field of communication? Describe a current or emerging trend. Identify the business need or communication problem the trend resolves. Explain the current or...
-
Provide two specific examples of how people are using or abusing mass media laws today. Consider the ethical implications as well the legal. Explain the laws that are being violated. Would you...
-
You're the human resources manager at the CSI Business Hotel in Chicago. You have an opening for a Guest Services Manager. Hoping for more customers by the summer, you know you will need to go all...
-
Evaluate the impact to the accounting profession should privately held companies be required to use U.S.GAAP while publicly traded companies are required to IFRS.
-
Q1) What is the a3 Value Q2) What is the a7 Value Q3) What is the a4 Value Q4) What is the b3 Value Q5) What is the b2 Value Q6) What is the sign of 2nd constraint? A pastry chef at a bakery wants to...
-
Runner???s gait. The amount of torque required by an external applied force (other than gravity) to move a leg forward is less than that required to move it backward if the leg is bent as shown in...
-
A wheel of mass 11 kg is pulled up a step by a horizontal rope as depicted in Figure P8.81. If the height of the step is equal to one-third the radius of the wheel, h = 1/3 R, what minimum tension is...
-
The end of a pencil of mass m and length L rests in a corner as the pencil makes an angle with the horizontal (x) direction (Fig. P8.82). If the pencil is released, it will rotate about point O with...
-
The Apple company is considering a machine to reduce operation costs. The machine costs $2B. If bought, the machine will reduce the cost of goods sold by $1B a year (before tax) for the next 3 years....
-
Explain why there is little adverse selection in Employer based health insurance programs, compared to the non-group private market for health insurance. 2. Suppose that in a country with a generous...
-
Mr Archie works for a basic wage of $650 per hour for a 40-hour week a)If Mr. Archie worked only 40 hours during a particular week, what was his wage for the week b) Mr. Archie's wage for the next...

Study smarter with the SolutionInn App


