Calculate the free energy change for this reaction at 25 C. Is the reaction spontaneous? 2 Ca(s)
Question:
Calculate the free energy change for this reaction at 25 °C. Is the reaction spontaneous?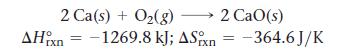
Transcribed Image Text:
2 Ca(s) + O2(g) — 2 CaO(s) −1269.8 kJ; Δ.Sixn -364.6J/K ΔΗ, xn
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
To calculate the free energy change G for the reaction we can use the equ...View the full answer

Answered By

Rayan Gilbert
I have been teaching since I started my graduation 3 years ago. As a student, working as Teacher/PA has been tough but made me learn the needs for student and how to help them resolve their problems efficiently. I feel good to be able to help out students because I'm passionate about teaching. My motto for teaching is to convey the knowledge I have to students in a way that makes them understand it without breaking a sweat.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Calculate the free energy change for this reaction at 25 C. Is the reaction spontaneous? C3H8(g) + 5O(g) 5 O(g) 3 CO(g) + 4HO(g) AHin = -2217 kJ; ASixn 101.1 J/K rxn
-
Calcium oxide, CaO, is manufactured by decomposition of calcium carbonate, CaCO3, in a furnace at 500 K: CaCO3(s)?CaO(s)+CO2(g) The spontaneity of a reaction can be determined from the free energy...
-
You monitor the reaction of a protein forming a dimer and find an apparent rate constants of 6 s-1 with 1 M total protein and 10 s-1 with 5 M total protein. a. What are the forward and reverse rate...
-
Explain how community service staff members can monitor the impact of work undertaken and/or services provided in line with the scope of their own work role?
-
The Internet accelerates the process of economic growth. Discuss.
-
Learn about the Securities and Exchange Commission (SEC). Address: www.sec.gov/index.html Step From the SEC homepage, choose About the SEC. Answer the following questions: (a) How many enforcement...
-
The accounting records of Dwter Minerals Corporation provide income statement data for 2008. Total expenses include depreciation of \(\$ 50,000\) computed under the straight-line method. In...
-
Myers Company Ltd. was formed 10 years ago by the issuance of 34,000 common shares to three shareholders. Four years later, the company went public and issued an additional 30,000 common shares. The...
-
1. Is the process by which correctional clients are selected discriminatory? 2. What might be done to reduce actual or perceived discrimination? Michael Santos served a very long sentence for his...
-
Fill in the blanks in the table. Both H and S refer to the system. T AS + AG Temperature dependent Low Temperature Spontaneous High Temperature Spontaneous Nonspontaneous Nonspontaneous
-
Calculate the change in Gibbs free energy for each of the sets of H rx n , S rx n , and T given in Problem 42. Predict whether or not each reaction is spontaneous at the temperature indicated....
-
In Exercises, find the area of the region bounded by the graphs of the equations. y = -x + x + 6, y = 0
-
Describe how the proposed change in procedure and practice impacted relevant stakeholders. Describe the behaviors of the team during implementation of an innovation team in an early childcare...
-
Hospitality in the Big City1. How does this case illustrate the importance of 2. How important is it to defuse unpleasantness even 3.Describe any similar experiences you may havehad where people...
-
Over the year, the Mars company has changed both the colors and the proportion of colors for M&M candies. The original colors were red, yellow, violet, green and brown. In 1995 blue was introduced....
-
Questions below based on figure above: Consider the Arithmetic Logic Unit (ALU) block diagram shown in Fig. 1(a). Each block is numbered in brackets, for example (1), (2)... (10). The D-Flip-Flops...
-
Inventory Beginning Balance Ending Balance $27,000 Accounts Receivables 21,000 Accounts Payables 10,000 $28,000 22,000 14,000 Consider the above balances and if Credit Sales were $175,000 and the...
-
At December 31, 2016, House Co. reported the following information on its balance sheet. Accounts receivable ............ $960,000 Less: Allowance for doubtful accounts .... 80,000 During 2017, the...
-
Q1) What is the a3 Value Q2) What is the a7 Value Q3) What is the a4 Value Q4) What is the b3 Value Q5) What is the b2 Value Q6) What is the sign of 2nd constraint? A pastry chef at a bakery wants to...
-
A chain drive (Fig. P8.77) transfers the rotational motion of a tractor transmission to a grain elevator. The drive shaft places a counterclockwise torque on the small sprocket of radius 2.2 cm so...
-
Bicycles like the one shown in the photo in Figure P8.78 typically have 24 possible gear ratios (three gears on the front and eight on the back). Suppose the diameters of the largest and smallest...
-
A bodybuilder is in training and performs curls with a 10-kg barbell to strengthen her biceps muscle. At the top of the stroke, her arm is configured as in Figure P8.79, with her forearm at 35 with...
-
Pepsico Inc. announced yesterday that it would spin off its KFC, Pizza Hut and Taco Bell restaurant businesses as a publicly traded company, one that would be second only to McDonald's in the fast...
-
Please follow the link and read the canvas project. Based on the reading, please answer the following questions . Link for the Startup Analysis Canvas Project:...
-
Your health facility, the Lakeland Medical Clinic was opened to serve area residents. The first two months of operation saw strong patient volume after which the numbers dropped off dramatically....

Study smarter with the SolutionInn App


