Consider the reaction: Each of the entries in the following table represents equilibrium partial pressures of A
Question:
Consider the reaction:
![]()
Each of the entries in the following table represents equilibrium partial pressures of A and B under different initial conditions.
What are the values of a and b in the reaction?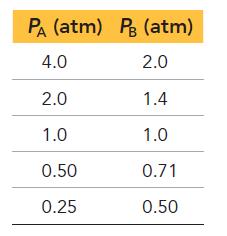
Transcribed Image Text:
aA(g) bB(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In Fig P2.40 the pressures at A and B are the same, 100 kPa. If water is introduced at A to increase pA to 130 kPa, find and sketch the new positions of the mercury menisci. The connecting tube is a...
-
You have been assigned the task of measuring the equilibrium constant for the reaction N 2 O 4 2NO 2 as a function of temperature. To do so, you evacuate a rigid 2-liter vessel equipped with a...
-
The equilibrium constant (KP) for the formation of the air pollutant nitric oxide (NO) in an automobile engine at 530°C is 2.9 Ã 10-11: (a) Calculate the partial pressure of NO under these...
-
Refer to the RMO CSMS Order Fulfillment subsystem shown in Figure. Draw a use case diagram that shows all actors and all use cases. Use a drawing tool such as Microsoft Visio if it is available.
-
Pittsburgh-Walsh Company (PWC) is a manufacturing company whose product line consists of lighting fixtures and electronic timing devices. The Lighting Fixtures Division assembles units for the...
-
A gasoline engine produces 20 hp using 35 Btu/s of heat transfer from burning fuel. What is its thermal efficiency and how much power is rejected to the ambient?
-
Know the reasons for using value management in projects.
-
The current sections of Sanford Inc.s balance sheets at December 31, 2013 and 2014, are presented here. Sanfords net income for 2014 was $153,000. Depreciation expense was $27,000. Instructions...
-
Consider the following WiFi topology, with five transmitters (A, B, D, E, and F) and two access points (C and G). As shown, A and B are associating with C, while D, E, and F are associating with G. 2...
-
A particular reaction has an equilibrium constant of K p = 0.50. A reaction mixture is prepared in which all the reactants and products are in their standard states. In which direction does the...
-
The reaction A(g) 2 B(g) has an equilibrium constant of K c = 1.0 at a given temperature. If a reaction vessel contains equal initial amounts (in moles) of A and B, does the direction in which the...
-
Fill out the following chart to show the impact on the accounting equation from each transaction. iablities Stockholders' equity Increase Decrease Increase Decrease Increase Decrease Assets Date...
-
Answer the following in a long paragraph form that is detailed and informative. LIFO Explain what LIFO is and its financial impacts. Why would some industries use LIFO over FIFO? What are the results...
-
share the influence your parent's Marine Corps or Navy service has had on you. Discuss what it means to you to know that your parent served in the Marine Corps or Navy. If your parent's service has...
-
If you were asked to justify the study of leadership, how would you do it? What is your leadership style? What do you see as your personal strengths and limitations when it comes to your leadership...
-
At least two economic trends which are impacting the H&M ' s operations ( e . g . , could be trends in demand and supply of their products / services , unemployment rate, consumer confidence in the...
-
Why is the Transfer price important to performance measurement? What precisely does the minimum transfer price attempt to compensate the selling division for? Why might an internal order cause the...
-
Ownership interests in a corporation are reported both in the balance sheet under shareholders' equity and in the statement of shareholders' equity. Required: a. List the principal transactions and...
-
What is the ideal number of children to have? This question was asked on the Sullivan Statistics Survey I. Draw a dot plot of the variable Children from theSullivanStatsSurveyI data set at...
-
Figure 9.27 shows a duct in which methyl alcohol at 25°C flows at the rate of 3000 L/min. Compute the energy loss over a 2.25-m length of the duct. All surfaces are smooth plastic. 100 mm 30 mm...
-
In Fig. 9.26, ethylene glycol (sg = 1.10) at 77°F flows around the tubes and inside the rectangular passage. Calculate the volume flow rate of ethylene glycol in gal/min required for the flow to...
-
Compute the energy loss for the flow of water in the cooling passage described in Problem 9.34 if its total length is 45 in. Use for steel. Also compute the pressure difference across the total...
-
2. Cindy runs 13 blocks to the park, and 13 blocks back home every day. If she does this 4 days a week, how many blocks will she run? Answer: Compute: blocks Explain:
-
3. Decompose the number below two ways. 2 1/2 2 11/12 4. Compare using >, < or =. 20 13 10 36 48 5. Rewrite the improper fraction as a mixed number. 85 10 23
-
(5pts) Let X1, X2 be a random sample of size n = 2 from the distribution having pdf f(x; 6) (1/0)e/0,0 < x < o, zero elsewhere. We reject Ho: 02 and accept H: 0 = 1 if the observed values of X1, X2,...

Study smarter with the SolutionInn App


