Consider the three generic weak acids HA, HB, and HC. The images shown here represent the ionization
Question:
Consider the three generic weak acids HA, HB, and HC. The images shown here represent the ionization of each acid at room temperature. Which acid has the largest Ka?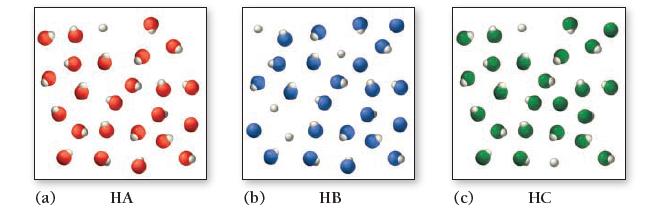
Transcribed Image Text:
(a) HA (b) HB (c) HC
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
b HB...View the full answer

Answered By

Douglas Makokha
Unlock Academic Success with Dedicated Tutoring and Expert Writing Support!
Are you ready to excel in your academics? Look no further! As a passionate tutor, I believe that dedication and hard work are the keys to achieving outstanding results. When it comes to academics, I strive to provide nothing but the best for every student I encounter.
With a relentless thirst for knowledge, I have extensively researched numerous subjects and topics, equipping myself with a treasure trove of answers to tackle any question that comes my way. With four years of invaluable experience, I have mastered the art of unraveling even the most intricate problems. Collaborating with esteemed writers has granted me exclusive access to the trade secrets utilized by the industry's top professionals.
Allow me the pleasure of assisting you with your writing assignments. I thrive on challenges and will guide you through any obstacles you may face. Together, we will unlock your academic potential and pave the way for your success.
4.90+
60+ Reviews
345+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
The diagrams here show three weak acids HA (A = X, Y, or Z) in solution. (a) Arrange the acids in order of increasing Ka. (b) Arrange the conjugate bases in increasing order of Kb. (c) Calculate the...
-
Consider the acids in Table. Which acid would be the best choice for preparing a pH = 7.00 buffer? Explain how to make 1.0 L of this buffer. Table Values of Ka for Some Common Monoprotio Acids...
-
Photons of wavelength 1.68 cm excite transitions from the rotational ground state to the first rotational excited state in a gas. Whats the rotational inertia of the gas molecules?
-
The theoretical cycle time for a product is 48 minutes per unit. The budgeted conversion costs for the manufacturing cell dedicated to the product are $4,320,000 per year. The total labor minutes...
-
Figure P14.16 shows Superman attempting to drink water through a very long straw. With his great strength he achieves maximum possible suction. The walls of the tubular straw do not collapse. (a)...
-
Explain the idea behind temporal logic.What kinds of problems is it useful for solving? Give three examples.
-
On March 7, 2012, Celgene Corporation acquired all of the outstanding stock of Avila Therapeutics, Inc. in exchange for $352.2 million in cash plus contingent consideration. Referring to Celgenes...
-
When light of wavelength 3.50x102 nm falls on a potassium surface, electrons having a maximum kinetic energy of 1.31 eV are emitted. a. Find the work function of potassium. b. Find the cutoff...
-
Consider the given acid ionization constants. Identify the strongest conjugate base. Acid HNO(aq) HCHO(aq) HCIO(aq) HCN(aq) a) NO (aq) c) CIO (aq) Ka 4.6 x 10-4 1.8 x 10-4 2.9 X 10 8 4.9 10-10 b)...
-
Calculate the pH of each solution at 25 C and indicate whether the solution is acidic or basic. (a) [H3O+] 1.8 x 10-4 M = (b) [OH-] = 1.3 x 10-2 M
-
What is meant by market power? What are the ways in which a monopoly gains market power?
-
Identify at least two plausible service departments in your industry* (not including HR, IT or maintenance). Describe the departments, the services they provide to the producing departments or to...
-
Watch this video, and then describe the role of balance sheets, income statements and statements of cashflows to companies. Pick a company off the Dow Industrial list Links to an external site.,...
-
Conduct research to find out one or more differences that exist between balance sheets that are prepared using U.S. GAAP and those prepared using International Financial Reporting Standards (IFRS)....
-
In 1988, Nandalal's family immigrated to Canada- he was 2 years old. His grandparents and parents pooled their money and invested in a plot of land located in the greater Vancouver area. They started...
-
Assume Kirtch Company drops Product C. The company then doubles the production and sales of Product B without increasing fixed costs. a. Fill in the chart below to use the total cost approach. After...
-
Why do managers sometimes manage earnings?
-
It is possible to investigate the thermo chemical properties of hydrocarbons with molecular modeling methods. (a) Use electronic structure software to predict cHo values for the alkanes methane...
-
The vision of a hyperope is corrected with a +9D spectacle lens worn 12 mm from the cornea. Determine the appropriate power of a replacement contact lens.
-
We wish to correct the vision of a 7D my ope, whose both eyes are the same, with spectacles worn 15 mm from the eye. Determine the appropriate power.
-
An optometrist finds that a farsighted person has a near point at 125 cm. What power will be required for contact lenses if they are effectively to move that point inward to a more workable distance...
-
1. Which statement by a nurse reflects an accurate understanding of systems theory? a.My patient has anemia reflected by low RBC, Hgb, and Hct values, so I have to monitor for fatigue, dyspnea, and...
-
Neo-institutional theory suggests that, in order to survive, organizations need to gain legitimacy and do so through isomorphism with salient institution. According to your understanding Provide a...
-
1. Select the scientific theory used in several nursing theories to explain the following scenario: One nurse, whose adult son died in the ER 11 years ago, contends that she can never walk into a...

Study smarter with the SolutionInn App


