Determine whether each reaction is an oxidationreduction reaction. For each oxidationreduction reaction, identify the oxidizing agent and
Question:
Determine whether each reaction is an oxidation–reduction reaction. For each oxidation–reduction reaction, identify the oxidizing agent and the reducing agent.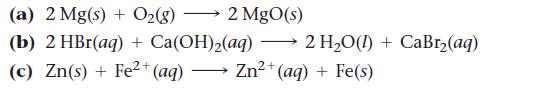
Transcribed Image Text:
(a) 2 Mg(s) + O₂(8) 2 MgO(s) (b) 2 HBr(aq) + Ca(OH)₂(aq) → 2 H₂O(I) + CaBr₂(aq) 2+ (c) Zn(s)+Fe²+ (aq) Zn²+ (aq) + Fe(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
This is a redox reaction because magnesium increases in o...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the redox reaction (a) Identify the oxidizing agent on the left side of the reaction and write a balanced oxidation half-reaction. (b) Identify the reducing agent of the left side of the...
-
(A) Is H 2 (g) an oxidizing or reducing agent in the reaction below? Explain. 2 NO 2 (g) + 7 H 2 (g) 2 NH 3 (g) + 4 H 2 O(g) (B) Identify the oxidizing agent and the reducing agent in the following...
-
Determine whether each reaction is a redox reaction. For each redox reaction, identify the oxidizing agent and the reducing agent. a. Al(s) + 3 Ag+ (aq) b. SO3(g) + HO(1) c. Ba(s) + Cl(g) d. Mg(s) +...
-
Consider an atom diffuses in a 3 D simple cubic lattice by a random walk mechanism. The atom jumps 6 x 1 0 - 5 times per second at 3 0 0 K and 3 x 1 0 4 times per second at 6 0 0 K . Assuming that...
-
Discuss the differences between traditional ERP and extended enterprise systems.
-
Show that the moment of inertia of a spherical shell of radius R and mass m is 2mR 2 /3. This can be done by direct integration or, more easily, by finding the increase in the moment of inertia of a...
-
Bernardino Santos-Rodiguez was piloting a boat owned by his friend, Raul Viera-Torres, in waters near Guayama, Puerto Rico. The boat was equipped with a hydraulic steering system manufactured by...
-
Norman Concrete Company pours concrete slabs for single-family dwellings. Wayne Construction Company, which operates outside Normans normal sales territory, asks Norman to pour 40 slabs for Waynes...
-
Thunderduck Shoes provides shoe shining and repair services to customers. For the year which ended Dec 31, the company reports the following amounts: Account Amount Account Amount Rent Expense 22,400...
-
What is a gas-evolution reaction? Give an example.
-
Explain the principles behind an acidbase titration. What is an indicator?
-
a. What forms and documents would KKD use in the process described? Which generic step(s) in the sales / collection process would use each form? b. In a manner specified by your instructor, document...
-
To differentiate between the various market segments, the firm must eliminate barriers that identify product or service attributes that the segments value differently. create barriers by identifying...
-
Which of the following is an approach that firms can use when managing inventory to meet predictable demand variability? Time flexibility from workforce Use of seasonal workforce Using common...
-
In what way do supply chain flows affect the success or failure of a firm such as Benetton? List two supply chain decisions that have a significant impact on supply chain profitability.
-
Which of these is a relative measurement? Tons Kilowatt-hours Parts per million Kelvin
-
The capacity management approach where a firm builds facilities to produce a relatively stable output of products over time in a very efficient manner, and facilities to produce a widely varying...
-
Pallest Corporation reported the following pretax information for its current fiscal year: $40,000 income from continuing operations and an $8,000 extraordinary loss. Pallest is subject to a 30%...
-
Cassandra Casey operates the Futuristic Antique Store. She maintains subsidiary ledgers for accounts payable and accounts receivable. She presents you with the following information for October 2019:...
-
You wish to replace the flooring in your kitchen with square linoleum tiles that measure 12 inches on a side. If your kitchen has a floor area of 10.7 square yards, how many such tiles will you need...
-
A typical human baby at birth is about 20 inches as measured from head to toe. An adult is much longer. What is the approximate ratio of the height of an adult female to the length of a baby at birth?
-
The solar system in perspective. The diameters of the Earth and the Sun are approximately 1.3 10 7 m and 1.4 10 9 m respectively, and the average SunEarth distance is 1.5 10 11 m. Consider a scale...
-
QUESTION 3: EXPLAIN WITH EXAMPLE WHAT IS THE COST POOL?
-
Describe a scenario where miscommunication led to a misunderstanding among team members. How could this situation have been avoided or resolved?
-
Analyze a case where a company's culture clash led to negative consequences. How did conflicting values or norms affect employee morale, teamwork, and overall productivity?

Study smarter with the SolutionInn App


