Determine whether each reaction is a redox reaction. For each redox reaction, identify the oxidizing agent and
Question:
Determine whether each reaction is a redox reaction. For each redox reaction, identify the oxidizing agent and the reducing agent.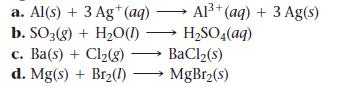
Transcribed Image Text:
a. Al(s) + 3 Ag+ (aq) b. SO3(g) + H₂O(1)→ c. Ba(s) + Cl₂(g) d. Mg(s) + Br₂(1) Al³+ (aq) + 3 Ag(s) H₂SO4(aq) → BaCl₂(s) MgBr₂(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a Als 3 Ag aq Al3aq 3 Ags This is a redox reaction The aluminum metal ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Determine whether each reaction is a redox reaction. For each redox reaction, identify the oxidizing agent and the reducing agent. a. 4 Li(s) + O(g) 2 LiO(s) b. Mg(s) + Fe+ (aq) Mg2+(aq) + Fe(s)...
-
Consider the redox reaction (a) Identify the oxidizing agent on the left side of the reaction and write a balanced oxidation half-reaction. (b) Identify the reducing agent of the left side of the...
-
stock options are no longer as valuable and employees in startups are losing out to founders and early investors for all of these reasons except: there is higher employee turnover, less of a delay in...
-
j) Assume that one of these portfolio's is the Market Portfolio and all portfolios, except Portfolio G, are fairly priced according to the CAPM. Derive the Treynor Measure for these fairly priced...
-
The Gayton Menswear company was founded by Fred Williams in 1986 and has grown steadily over the years. Fred now has 17 stores located throughout the central and northern parts of the state. Because...
-
Cheryl Montoya picked up the phone and called her boss, Wes Chan, the vice president of marketing at Piedmont Fasteners Corporation: Wes, Im not sure how to go about answering the questions that came...
-
Continuing to focus on evidence associated with the act, concealment, and conversion, use the evidentiary material to continue the examination. In addition, the examiner also starts to think of terms...
-
Perdue Company purchased equipment on April 1, 2012, for $270,000. The equipment was expected to have a useful life of three years, or 18,000 operating hours, and a residual value of $9,000. The...
-
A figure skater spins at the end of her routine and slows down with an angular acceleration of "0.4" A per second squared. If she initially spun with a frequency of 2.35 Hz, how much time does it...
-
Determine whether each redox reaction occurs spontaneously in the forward direction. a. Ni(s) + Zn+ (aq) b. Ni(s) + Pb+ (aq) c. Al(s) + 3 Ag+ (aq) d. Pb(s) + Mn+ (aq) 2+ Ni+(aq) + Zn(s) Ni+(aq) +...
-
What is the oxidation state of Cl in each ion? a. CIO- b. ClO c. ClO3 d. CIO4
-
For the three-cart system illustrated in Figure AP2.5, obtain the equations of motion. The system has three inputs u1, u2, and u3 and three outputs x1, x2, and x3. Obtain three second-order ordinary...
-
Read those clauses of the general conditions of the contract for construction that refer to the owner, architect, contractor, and subcontractor. Then list the major responsibilities of these...
-
Alright Construction, Inc., signs a contract with Casa Del Monte, Inc., to build five new 20-room modules for a large tourist resort in a Caribbean country. Casa Del Monte establishes liquidated...
-
Spotless Dry Cleaners had these balances in their ledger accounts on 31 March 20x7. You are required to: 1. Prepare a trial balance in accordance with Figure 6.5. 2. Prepare the financial statements...
-
In Exercise 33.31, is there an ask where Venezuelans may say no thank you to trading with Canada? Exercise 33.31 Review the numbers for Canada and Venezuela from Table 33.12 which describes how many...
-
Refer to Exhibit 9.5 (pg. 325). For each pair of factors, describe why you believe the factor in the right column provides a higher level of assurance. That is, why do these factors indicate a...
-
Refer to RE7-9. On December 31, Jordan, Inc., received $50,000 on assigned accounts. Prepare Jordan, Inc.s, journal entries.
-
Element compound homogeneous mixture (heterogeneous mixture) 4) A piece of gold has a mass of 49.75 g. What should the volume be if it is pure gold? Gold has a density of 19.3 g/cm (3 points) D=m/v...
-
The viscosity of an oil is given as 80 SUS at 100F. Determine the viscosity in m 2 /s.
-
Convert a viscosity measurement of 6.5 10 -3 Pas into the units of lbs/ft 2 .
-
An oil container indicates that it has a viscosity of 0.12 poise at 60C. Which oil in Appendix D has a similar viscosity?
-
How to revised this words This method, along with "Alternate day", "5:2" and "36 hour fast" are considered the long period fasts.
-
How do you determine the market value of a publicly traded company? write a brief procedure.
-
Can you think of any specific components that are essential to include in budget analysis and financial planning?

Study smarter with the SolutionInn App


