Determine whether each redox reaction occurs spontaneously in the forward direction. a. Ca+ (aq) + Zn(s) b.
Question:
Determine whether each redox reaction occurs spontaneously in the forward direction.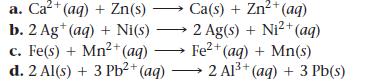
Transcribed Image Text:
a. Ca²+ (aq) + Zn(s) b. 2 Ag+ (aq) + Ni(s) c. Fe(s) + Mn²+ (aq) d. 2 Al(s) + 3 Pb²+ (aq) Ca(s) + Zn²+ (aq) 2 Ag(s) + Ni²+ (aq) Fe²+ (aq) + Mn(s) 2 Al³+ (aq) + 3 Pb(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a Ca2 aq Zns Cas Znaq The halfreactions involved are Zns Znaq 2e E 076 V Caaq 2e Cas E 287 V Since t...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Determine whether each redox reaction occurs spontaneously in the forward direction. a. Ni(s) + Zn+ (aq) b. Ni(s) + Pb+ (aq) c. Al(s) + 3 Ag+ (aq) d. Pb(s) + Mn+ (aq) 2+ Ni+(aq) + Zn(s) Ni+(aq) +...
-
At 298 K, for the reaction 2 PCl 3 (g) + O 2 (g) 2 POCl 3 (l), r H = -620.2 kJ mol -1 and the standard molar entropies, in J mol 1 K 1 , are PCl 3 (g), 311.8; O 2 (g), 205.1; and POCl 3 (l), 222.4....
-
At 298 K, for the reaction 2 H + (aq) + 2 Br - (aq) + 2 NO 2 (g) Br 2 (l) + 2 HNO 2 (aq), r H = -61.6 kJ mol -1 and the standard molar entropies are H + (aq), 0 J mol -1 K -1 ; Br - (aq), 82.4 J...
-
In a bygone day, airlines issued discount tickets to students who would be willing to fly on a particular day, with no notice, at a discounted price, one needed to show proof of being of student. The...
-
As a recently hired internal auditor for the Emerson Department Store (which has approximately 500 employees on its payroll), you are currently reviewing the stores procedures for preparing and...
-
Northwood Company manufactures basketballs. The company has a ball that sells for $25. At present, the ball is manufactured in a small plant that relies heavily on direct labor workers. Thus,...
-
Quilts R Us (QRU) is considering investing in a new patterning attachment with the cash flow profile shown in the table below. QRU's MARR is 13.5 percent/year. a. What is this investment's external...
-
The trial balance of Avtar Sandhu Co. shown below does not balance. Each of the listed accounts has a normal balance per the general ledger. An examination of the ledger and journal reveals the...
-
Would you make any changes to the Federal tax policy related to this area of the IRC ? If so , what would you change and why? How would these changes help rectify the disparate outcomes by race?
-
Suppose you wanted to cause Ni 2+ ions to come out of solution as solid Ni. Which metal could you use to accomplish this?
-
Determine whether each reaction is a redox reaction. For each redox reaction, identify the oxidizing agent and the reducing agent. a. Al(s) + 3 Ag+ (aq) b. SO3(g) + HO(1) c. Ba(s) + Cl(g) d. Mg(s) +...
-
Are there any other factors that need to be considered in a lease versus borrow and buy decision of this type? Explain.
-
Which of the following statements about U.S. production is correct? A. Government goods and services and export goods and services each account for the same percentage of total production. B. Capital...
-
Choosing among renting, leasing, and purchasing an AIS is strictly a financial decision and should be done by the finance staff. Do you agree? Discuss fully.
-
What role does each HR manager listed in Figure 14.2 (pg. 526) play? FIGURE 14.2 Organization Chart of the HR Function Manager of safety, health services, and benefits Manager of employee and...
-
Visit a local building contractor and determine how he or she handles control of submittals from subcontractors to architect/engineer. What system does he or she use to ensure the job will not be...
-
Determine the local prices for some bulk materials such as concrete, sand, cement, steel mesh, bricks, and lumber and compare them to the periodically published prices in Engineering News Record.
-
Kaseys Cake Shop made $20,000 in sales of wedding cakes in July. All of these sales were on bank credit cards. The credit card company charges a 3.5% collection fee. Prepare Kaseys journal entry to...
-
Why should you not model a decision variable as a random variable with a probability distribution?
-
Two hockey players are traveling at velocities of v 1 = 12 m/s and v 2 = -18 m/s when they undergo a head-on collision. After the collision, they grab each other and slide away together with a...
-
Consider an elastic collision in one dimension that involves objects of mass 2.5 kg and 4.5 kg. The larger mass is initially at rest, and the smaller one has an initial velocity of 12 m/s. Find the...
-
Two billiard balls undergo an elastic collision as shown in Figure P7.27. Ball 1 is initially traveling along x with a speed of 10 m/s, and ball 2 is at rest. After the collision, ball 1 moves away...
-
submit typed answer in good format with supporting calculations:uncertainty its operates under ideal conditions of uncertainty, with a fiscal year end of December 31st. its cash flows depend...
-
What SEC issues related to accounting data and operations and remedial actions assigned and penalties are given to Ixis Victor Alston, CEO of Ixis. Provide sources an date.
-
Define the accrued liabilities related to accrued commissions. write a brief intro on accured liabilities.

Study smarter with the SolutionInn App


