Determine whether each redox reaction occurs spontaneously in the forward direction. a. Ni(s) + Zn+ (aq) b.
Question:
Determine whether each redox reaction occurs spontaneously in the forward direction.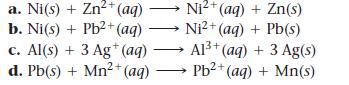
Transcribed Image Text:
a. Ni(s) + Zn²+ (aq) b. Ni(s) + Pb²+ (aq) c. Al(s) + 3 Ag+ (aq) d. Pb(s) + Mn²+ (aq) 2+ Ni²+(aq) + Zn(s) Ni²+(aq) + Pb(s) Al³+ (aq) + 3 Ag(s) Pb²+ (aq) + Mn(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
b and c oc...View the full answer

Answered By

Vijesh J
My passion to become a tutor is a lifetime milestone. Being a finance and marketing professional with hands-on experience in wealth management, portfolio management, team handling and actively contributing in promoting the company. Highly talented in managing and educating students in most attractive ways were students get involved. I will always give perfection to my works. Time is the most important for the works and I provide every answer on time without a delay. I will proofread each and every work and will deliver a with more perfection.
4.70+
5+ Reviews
15+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Determine whether each redox reaction occurs spontaneously in the forward direction. a. Ca+ (aq) + Zn(s) b. 2 Ag+ (aq) + Ni(s) c. Fe(s) + Mn+ (aq) d. 2 Al(s) + 3 Pb+ (aq) Ca(s) + Zn+ (aq) 2 Ag(s) +...
-
At 298 K, for the reaction 2 PCl 3 (g) + O 2 (g) 2 POCl 3 (l), r H = -620.2 kJ mol -1 and the standard molar entropies, in J mol 1 K 1 , are PCl 3 (g), 311.8; O 2 (g), 205.1; and POCl 3 (l), 222.4....
-
At 298 K, for the reaction 2 H + (aq) + 2 Br - (aq) + 2 NO 2 (g) Br 2 (l) + 2 HNO 2 (aq), r H = -61.6 kJ mol -1 and the standard molar entropies are H + (aq), 0 J mol -1 K -1 ; Br - (aq), 82.4 J...
-
To some U.S. manufacturers and labor unions, a cheap yuan value gives China's advantage in the global economy. bankers exports importers subsidies an unfair
-
The Cuts-n-Curves Athletic Club is a state-wide chain of full-service fitness clubs that cater to the demographics of the state (about 60% of all adults are single). The clubs each have an indoor...
-
Provide some examples of front-of-the-house and back-of-the-house service operations.
-
If a company with a 12/31 year-end lends money in the form of a 6-month note on 11/1, which accounts will be credited when the note is paid off on 4/30?
-
A negotiable promissory note executed and delivered by B to C passed in due course and was indorsed in blank by C, D, E, and F. G, the present holder, strikes out Ds indorsement. What is the...
-
An epicyclic gear train consists of a sun wheel S, a stationary annular wheel E and three identical planetwheels P carried on a star-shaped carrier C, as shown in Fig.15.14. The size of different...
-
Suppose you wanted to cause Ni 2+ ions to come out of solution as solid Ni. Which metal could you use to accomplish this?
-
Determine whether each reaction is a redox reaction. For each redox reaction, identify the oxidizing agent and the reducing agent. a. Al(s) + 3 Ag+ (aq) b. SO3(g) + HO(1) c. Ba(s) + Cl(g) d. Mg(s) +...
-
Calculate the value of a six-month futures contract on a Treasury bond. You have the following information: Six-month interest rate: 10% per year, or 4.9% for six months. Spot price of bond: 95. ...
-
The position vs. time graph below is for an object that is exhibiting SHM. The position function is: x(t) = Acos(at+D). Use this graph to fill in the table. x (m) AA 10 15 20 25 20 25 30 35 40 25 -25...
-
How would you make l a Personal Marketing Plan to market yourself? Give an example: In this situation, you consider yourself to be a "product" and develop a marketing plan to promote yourself career...
-
1) All resistors in the circuit shown have a resistance of 2.0 2. What is the equivalent resistance seen by the voltage source (in (2)? a. 0.80 b. 2.0 C. 3.0 d. 3.3 e. 5.0 2+2=4 4//2=4/3 2+1.3=3.3 B...
-
Duke Blue Devil Inc. just paid a dividend of $ 2 . 0 0 . Its stock is now selling for $ 4 8 per share. The firm is 5 0 % as risky as the market. The expected return on the market is 1 4 % , and the...
-
A flying squid ( family Ommastrephidae ) is able to "jump" off the surface of the sea by taking water into its body cavity and then ejecting the water vertically downward. A 0 . 7 6 - kg squid is...
-
Consider the following three bonds with $1,000 face value: Bond A: 10-year, 10 percent coupon bond Bond B: 10-year, zero-coupon bond Bond C: 20-year, 10 percent coupon bond Compute the market values...
-
Element compound homogeneous mixture (heterogeneous mixture) 4) A piece of gold has a mass of 49.75 g. What should the volume be if it is pure gold? Gold has a density of 19.3 g/cm (3 points) D=m/v...
-
A baseball pitcher (m = 80 kg) is initially standing at rest on an extremely slippery, icy surface. He then throws a baseball (m = 0.14 kg) with a horizontal velocity of 50 m/s. What is the recoil...
-
Two cars collide head-on and lock bumpers on an icy (frictionless) road. One car has a mass of 800 kg and an initial speed of 12 m/s and is moving toward the north. The other car has a mass of 1200...
-
Two cars of equal mass are traveling as shown in Figure P7.30 just before undergoing a collision. Before the collision, one of the cars has a speed of 18 m/s along +x and the other has a speed of 25...
-
In an effort to keep low-wage workers' salaries commensurate with the cost of living, a number of states have amended their constitutions to allow the minimum wage to be adjusted with inflation. You...
-
On January 1, 2024, a company adopted the dollar-value LIFO method. The inventory value for its one inventory pool on this date was $400,000. Inventory data for 2024 through 2026 are as follows: Date...
-
Are there different types of research used to identify market segments? If there are, can you provide three types of research that are used to identify market segments? Explain with the help of...

Study smarter with the SolutionInn App


