For each of the reactions, calculate the mass (in grams) of the product that forms when 3.67
Question:
For each of the reactions, calculate the mass (in grams) of the product that forms when 3.67 g of the underlined reactant completely reacts. Assume that there is more than enough of the other reactant.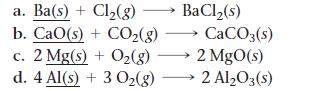
Transcribed Image Text:
a. Ba(s) + Cl₂(g) b. CaO(s) + COz(g) CaCO3(s) c. 2 Mg(s) + O₂(g) →→→ 2 MgO(s) d. 4 Al(s) + 3 O₂(g) 2 Al₂O3(s) BaCl₂(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a 556 g Ba...View the full answer

Answered By

SILPA MARY THOMAS
I have done my graduation and post-graduation in Chemistry. I have one year research experience and 4+ years teaching experience.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the reactions, calculate the mass (in grams) of the product that forms when 15.39 g of the underlined reactant completely reacts. Assume that there is more than enough of the other...
-
1. How strong are the competitive forces confronting J. Crew in the market for specialty retail? Do a [Michael Porter] five-forces analysis to support your answer. (see chapter 3 in the textfor...
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
Hernandez Company began 2010 with a $120,000 balance in retained earnings. During the year, the following events occurred: 1. The company earned net income of $80,000. 2. A material error in net...
-
Romo, Inc., has current assets of $1,850, has current assets of $1,850, net fixed assets of $8,600, current liabilities of $1,600, and long-term debt of $6,100. What is the value of the shareholders...
-
The Schultz Company prepares interim financial statements at the end of each quarter. The income statement presented at the end of the first quarter of 2007 is as follows: Shown next is the Schultz...
-
What is the purpose of a declaration?
-
a. For each of the four columns of the table, classify the data as either qualitative or quantitative; if quantitative, further classify it as discrete or continuous. Also identify the variable under...
-
Suppose a NTT company bond, maturing in one year, can be purchased today for $975. Assuming the bond is held to maturity, the investor will receive $1000 (principal) plus 7% interest (that is .07 x...
-
You have recently become the CFO for Beta Manufacturing, a small cap company that produces auto parts. As you step into your new position, you have decided to compile a report that details all...
-
Find the limiting reactant for each initial amount of reactants. a. 2 mol Na, 2 mol Br 2 b. 1.8 mol Na, 1.4 mol Br 2 c. 2.5 mol Na, 1 mol Br 2 d. 12.6 mol Na, 6.9 mol Br 2 2 Na(s) + Br(g) Br(g) 2...
-
Sulfuric acid dissolves aluminum metal according to the reaction: Suppose you want to dissolve an aluminum block with a mass of 15.2 g. What minimum mass of H 2 SO 4 (in g) do you need? What mass of...
-
Indicate what impact the following transactions would have on the accounting equation, Assets = Liabilities + Equity. A. B. C. D. Received cash from issuance of common stock Sold goods to customers...
-
Describe circumstances under which you would use a combination.
-
A committee of eight people must choose a president, a vice president, and a secretary. In how many ways can this be done?
-
A college student is chosen at random. Event A is that the student is an only child, and event B is that the student has a brother. Are events A and B mutually exclusive?
-
A pollster plans to sample 1500 voters from a city in which there are 1 million voters. Can the sampled voters be treated as independent? Explain.
-
A penny and a nickel are tossed. Each is a fair coin, which means that heads and tails are equally likely. a. Construct a sample space containing equally likely outcomes. Each outcome should specify...
-
Early in the chapter, we argued that "every organization must make a product or provide a service that someone values." Can you think of an example in which poor operations or supply chain management...
-
Explain why each of the following is either a private good or a public good: traffic lights, in line skates, a city park, a chicken salad sandwich, a tennis racket, national defense, a coastal...
-
When 2-ethyl-5-chlorotoluene was treated with sodium hydroxide at high temperature, followed by treatment with H 3 O + , three constitutional isomers with molecular formula C 9 H 12 O were obtained....
-
You wish to design an effusion source for Br atoms from Br 2 (g). If the source is to operate at a total pressure of 7.5 Torr, what temperature is required to produce a degree of dissociation of...
-
Calculate G for the isothermal expansion of 2.25 mol of an ideal gas at 325 K from an initial pressure of 12.0 bar to a final pressure of 2.5 bar.
-
Needham Pharmaceuticals has a profit margin of 4% and an equity multiplier of 1.8. Its sales are $130 million and it has total assets of $60 million. What is its return on equity (ROE)? Do not round...
-
Oriole Corporation manufactures safes-large mobile safes, and large walk-in stationary bank safes. As part of its annual budgeting process, Oriole is analyzing the profitability of its two products....
-
On the following graph, use the blue points (circle symbol) to plot Simone's initial production possibilities frontier (PPF). EMBROIDERED SWEATERS 30 25 8 15 10 5 0 0 1 2 3 4 TEA TOWELS 5 00 7 8...

Study smarter with the SolutionInn App


