List all the possible products for each alkane substitution reaction. a. CH3CH3 + Br b. CH3CHCH3 +
Question:
List all the possible products for each alkane substitution reaction.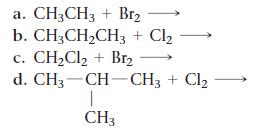
Transcribed Image Text:
a. CH3CH3 + Br₂ b. CH3CH₂CH3 + Cl₂ c. CH₂Cl₂ + Br₂ d. CH3-CH-CH3 + Cl₂ 1 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
a CH3CH Br b C...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
List all the possible products for each alkane substitution reaction. a. CH4 + Cl b. CH3CHBr + Br C. CH3CHCHCH3 + Cl d. CH3CHBr2 + Br
-
List all the possible bonds that can occur between the elements P, Cs, O, and H. Predict the type of bond (ionic, covalent, or polar covalent) one would expect to form for each bond.
-
List all the possible subshells and orbitals associated with the principal quantum number n, if n = 6.
-
In September 2020 Kevin sells a drawing for 2,000. He bought the drawing in February 2014 for 50,000 when it was thought (incorrectly) to be by a famous artist. Compute the allowable loss.
-
Emma Louise Company had net sales of $2,225,500,000 and cost of goods sold of $1,557,850,000. Emma had the following balances: Required: Round answers to two decimal places. 1. Calculate the average...
-
Mary is considering opening a new grocery store in town. She is evaluating three sites: downtown, the mall, and out at the busy traffic circle. Mary calculated the value of successful stores at these...
-
A project has been selected for implementation. The net cash flow (NCF) profile associated with the project is shown below. MARR is 10 percent/year. a. What is the internal rate of return of this...
-
Denny Asbestos Removal Company removes potentially toxic asbestos insulation and related products from buildings. The companys estimator has been involved in a long-simmering dispute with the on-site...
-
Part A An oxygen atom at a particular site within a DNA molecule can be made to execute simple harmonic motion when illuminated by infrared light. The oxygen atom is bound with a spring-like chemical...
-
Name each alkene. a. CH,=CHCH,CH, CH3 CH3 T T CH3-CH-C=CH-CH3 CH,=HCCHCH,CH, CH3 b. c. CH3 CH I CH3 CH3 I d. CH3-CH-CH=C-CH3 CH - CH3
-
Complete and balance each hydrocarbon combustion reaction. a. CH,CH,CH,CH, + O, b. CH=CHCH3 + 02 c. CH=CCHCH3 + O
-
The stockholders' equity section of Carey Co.'s balance sheet at December 31, 2010, was as follows: Common stock--$10 par (authorized 1,000,000 shares, issued and outstanding 600,000 shares) $...
-
3. Using the Internet, choose a country with a credit rating from Standard & Poor's that lies between "B" and "BBB" (inclusive, meaning the country may also be rated either B or BBB) and determine...
-
This is the bigger picture. The entirety of this process is the point in the new hire's career that you build a bond between the organization and the employee. This statement what describes?
-
Juan is a citizen and resident of Brazil. During the current year, Juan never visits the United States, nor does he hold a green card. However, he received a dividend from Macro Corporation, received...
-
This week's reading explores the way in which the structure of an organization can influence how projects are managed. For your initial post, introduce yourself to your classmates and identify an...
-
A lumber company is predicting annual lumber sales revenue based on the number of new home starts in the local economy. Using the estimated equation shown below, what is the forecasted number of...
-
Consider the types of poor driving habits that might occur at a traffic light. Make a list of the 10 you consider most likely to happen. Add the category of other to that list. (a) Compose a check...
-
The vapor pressure of the liquid NH, is measured at different temperatures. The following vapor pressure data are obtained. Temperature, K P, mmHg 217.1 223.4 234.7 588.1 Calculate the enthalpy of...
-
An electric field points into the page and occupies a circular region of radius 1.0 m, as shown in Fig. 29.14. There are no electric charges in the region, but there is a magnetic field forming...
-
The medical profession divides the ultraviolet region of the electromagnetic spectrum into three bands: UVA (320 nm420 nm), UVB (290 nm320 nm), and UVC (100 nm290 nm). UVA and UVB promote skin cancer...
-
Dielectric breakdown in air occurs when the electric field is approximately 3 MV/m. What would be the peak magnetic field in an electromagnetic wave with this peak electric field?
-
Critically critique your reading assignment for this module and point out any types of bias you may discern. Identify what type of bias and modify the statement(s) to show how you would present the...
-
Misra Inc. forecasts a free cash flow of $50 million in Year 3, ie, at t=3, and it expects FCF to grow at a constant rate of 5.5% thereafter. If the weighted average cost of capital (WACC) is 9.5%...
-
Presentation Outline - Instructions & Rubric make a detailed outline for your PowerPoint presentation. You may write it in point-form if you wish, but ensure that your point form notes are clear and...

Study smarter with the SolutionInn App


