Pick an appropriate solvent from Table 14.3 to dissolve each substance. State the kind of intermolecular forces
Question:
Pick an appropriate solvent from Table 14.3 to dissolve each substance. State the kind of intermolecular forces that would occur between the solute and solvent in each case.
a. Isopropyl alcohol (polar, contains an OH group)
b. Sodium chloride (ionic)
c. Vegetable oil (nonpolar)
d. Sodium nitrate (ionic)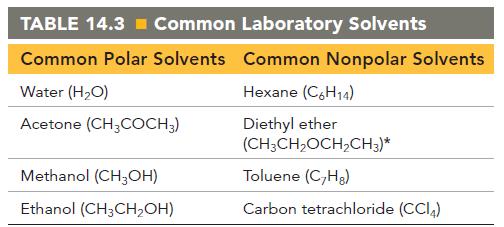
Transcribed Image Text:
TABLE 14.3= Common Laboratory Solvents Common Polar Solvents Common Nonpolar Solvents Hexane (C6H14) Diethyl ether (CH3CH₂OCH₂CH3)* Toluene (C₂Hg) Carbon tetrachloride (CC14) Water (H₂O) Acetone (CH3COCH3) Methanol (CH3OH) Ethanol (CH3CH₂OH)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a Isopropyl alcohol polar contains an OH group Appropriate solvent Water H2O or ethanol CHCHOH Inter...View the full answer

Answered By

Gilbert Chesire
I am a diligent writer who understands the writing conventions used in the industry and with the expertise to produce high quality papers at all times. I love to write plagiarism free work with which the grammar flows perfectly. I write both academics and articles with a lot of enthusiasm. I am always determined to put the interests of my customers before mine so as to build a cohesive environment where we can benefit from each other. I value all my clients and I pay them back by delivering the quality of work they yearn to get.
4.80+
14+ Reviews
49+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Pick an appropriate solvent from Table 14.3 to dissolve each substance. State the kind of intermolecular forces that would occur between the solute and solvent in each case. a. Motor oil (nonpolar)...
-
A.) Pick an appropriate solvent to dissolve acetic acid (polar,contains an OH group) . Water (H 2 O) Acetone (CH 3 COCH 3 ) Methanol (CH 3 OH) Ethanol (CH 3 CH 2 OH) Hexane (C 6 H 14 ) Diethyl...
-
For each compound, would you expect greater solubility in water or in hexane? Indicate the kinds of intermolecular forces that would occur between the solute and the solvent in which the molecule is...
-
Daniel and Karen Chapman have three children, aged 2, 8 and 11 at the end of the year. The 8 year old is blind and therefore qualifies for the disability tax credit. The other two are in good mental...
-
Refer to Exercise 12-9. Suppose that for 2010, McCawl Company has chosen suppliers that provide higher-quality parts and redesigned its plant layout to reduce material movement. Additionally, McCawl...
-
Andreka Inc. uses the direct method to allocate support department costs to production departments (fabricating and finishing). Information for June 2010 follows. a. What amounts of human resources...
-
Examine the suggestion that small and medium-sized companies do not need information from a core set of ecological indicators related to their business.
-
The following information is available for Pacilio Security Services Inc. for 2011, its first year of operations. Pacilio provides security services for local sporting events. The following summary...
-
Flounder Company began operations on January 1, 2019, adopting the conventional retail inventory system. None of the company's merchandise was marked down in 2019 and, because there was no beginning...
-
Which molecule would you expect to be more soluble in water: CH 3 CH 2 CH 2 OH or HOCH 2 CH 2 CH 2 OH?
-
What keeps the particles in a colloidal dispersion from coalescing?
-
Trace each step in the pathway from an olfactory receptor to the interpreting center of the cerebrum.
-
We want to increase the temperature of the air contained in a closed bottle. The initial temperature and pressure of the air inside the bottle are 300 K and 202,650 Pa absolute, respectively. The...
-
Tel Tec has just announced that their next dividend will be $2.25. In the announcement, management has projected that earnings will grow at 10 percent per year for the following 3 years, then slow to...
-
Investigate the molecular basis of autophagy, a highly conserved cellular process involved in the degradation and recycling of cellular components, and discuss its multifaceted roles in cellular...
-
I am IDENTITY AWARENESS ACTIVITY I am I am I am I am I am I am I am I am I am I am 1. How did you learn about this identity? When you learned this, how did you feel? 2. What are the advantages and...
-
What is FTX? How does it operate? What factors triggered its bankruptcy? What are the implications for cryptocurrencies at large?
-
Access the 2007 10-K for Abercrombie & Fitch (www .abercrombie.com). Required: (a) Compute the following ratios for Abercrombie & Fitch: (1) Current ratio (2) Quick ratio (3) Debt to total assets (4)...
-
Complete problem P10-21 using ASPE. Data from P10-21 Original cost ................................................................. $7,000,000 Accumulated depreciation...
-
A grand piano with three legs has a mass of 350 kg and is at rest on a level floor. (a) Draw a free-body diagram for the piano. Show the force of the floor on each leg as a separate force in your...
-
Your friends car has broken down, and you volunteer to push it to the nearest repair shop, which is 2.0 km away. You carefully move your car so that the bumpers of the two cars are in contact and...
-
A tall strongman of mass m = 95 kg stands upon a scale while at the same time pushing on the ceiling in a small room. Draw a free-body diagram of the strongman (Fig. P3.34) and indicate all normal...
-
Zboy Corp is liquidating. Upon liquidation, it will distribute the assets to its two shareholders, Lee (40% shareholder) and Kelly (60% shareholder). The assets have a FMV of $500,000 and an adjusted...
-
1. A dime is accidentally dropped from the top of the Empire State Building in Manhattan, New York. The height, h, in meters of the dime above the ground at t seconds can be modelled by 2 h(t)=- 4.9...
-
A deuteron particle (the nucleus of an isotope of hydrogen consisting of one proton and one neutron and having a mass of 3.3410-27 kg) moving horizontally enters a uniform, vertical, 0.640 T magnetic...

Study smarter with the SolutionInn App


