Pick the more oxidized structure in each pair. || a. CH3 CH or CH3-CH-OH b. CH3 -CH-OH
Question:
Pick the more oxidized structure in each pair.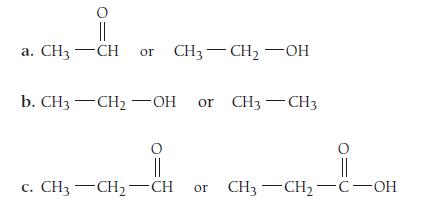
Transcribed Image Text:
|| a. CH3 CH or CH3-CH₂-OH b. CH3 -CH₂-OH or CH3-CH3 || c. CH—CH, CH or CH3CH₂C-OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
CH3CH2OH is more oxidized Oxidation is the loss of electronsso the more oxidiz...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
When discussing weighing schemes for calculating the weighted average cost of capital, ________. Question 10 options: book value weights and market value weights lead to the same value for the cost...
-
Expand the function ( f(x) = \ln(1+x) ) into a Taylor series around ( a = 0 ). Determine the radius of convergence for the series.
-
Can someone please summarize the case study below: if you know about the case IKEA Looks to Further Penetrate the U.S. Market 10 CASE Synopsis: IKEA is known around the world for its stylish,...
-
Provide a brief description of the JDBC URL.
-
Golding's finishing department had the following data for the month of July: Required: 1. Calculate unit costs for the following categories: transferred-in, materials, and conversion. 2. Calculate...
-
In order for her to get the data she needs, people will have to make comments on Sarahs film-tourism blog. How will Sarah ensure that people know about and find her blog so that they can keep posting...
-
Tennessee law imposes durational-residency requirements on persons and companies wishing to operate retail liquor stores, requiring applicants for an initial license to have resided in the state for...
-
This problem is based on the 2008 annual report of Intel Corporation in the appendix. Find in the Selected Financial Data (also known as the Five-Year Financial Summary), or calculate, the following...
-
One way financial managers evaluate a firm's current financial condition is by computing ratios based on current accounts listed on the firm's financial statements. Financial managers look at four...
-
Consider molecules that have two carbons and two chlorines. Draw the structures of three of these with no dipole moment and two with a dipole moment.
-
There are two compounds with the formula C 3 H 6 , one of which does not have a multiple bond. Draw its structure and explain why it is much less stable than the isomer with the double bond.
-
Find theLaplace transform of the function f(t) = t n e at , where n is an integer.
-
Comparative statements of financial positions for Mangrove Ltd. appear below: Mangrove Ltd. Statement of Financial Position Year Ended December 31, 2021 31-Dec-21 Assets Cash Accounts receivable...
-
Lundy Chemical Company produces a variety of products. Traditionally, the company has established base pricing for its products as 200% of the full manufacturing cost. However, one...
-
Why does the relationship between dependencies and interdependencies needs to be addressed and considered to preserve our national security and economic success, and with this condition, how does...
-
Consider an infinitely long circular cylinder with finite radius R and thermal conductivity k. There is a line-shaped heat source that generates heat at a rate of q, per unit axial length. This heat...
-
-1 0 A = 2 2 1 -2 (a) Find a basis for the kernel of the matrix 309 206 (b) For which values of a and b is the following matrix invertible? Show your work. 1 1 1 200 b b 338 a
-
You are attempting to develop a quality monitoring system for some parts purchased from Charles Sax Manufacturing Co. These parts are either good or defective. You have decided to take a sample of...
-
A copper sphere of 10-mm diameter, initially at a prescribed elevated temperature T;, is quenched in a saturated (1 atm) water bath. Using the lumped capacitance method, estimate the time for the...
-
An electrochemical impulse traveling along the cell modeled in Fig. 25.41 changes the value of 3 so now it supplies a 40-nA upward current. Assuming the rest of the circuit remains as described in...
-
In Problem 60, take C 1 = C 2 = C, and find the current through R 2 as a function of time. Use the node and loop laws to get a differential equation for the current, and use the initial conditions on...
-
In the circuit of Fig. 25.42 the switch is initially open and the capacitor is uncharged. Find expressions for the current I supplied by the battery (a) just after the switch is closed and (b) a long...
-
What would be an effective hedge ? A U.S. firm holds an asset in Great Britain and faces the following scenario: Probability Spot rate State 1 25% $ 2.20/ State 2 50% $ 2.00/ State 3 25% $ 1.80/E P'...
-
Poor communication will lead to demotivated staff and a lack of overall direction for the organization. Discuss.
-
What markets do you see as a good fit for Tim Hortons future expansion? Explain your choice

Study smarter with the SolutionInn App


