Recall that Cr and Cu are exceptions to the normal orbital filling, resulting in a [Ar] 4s
Question:
Recall that Cr and Cu are exceptions to the normal orbital filling, resulting in a [Ar] 4s13dx configuration.
Write the ground state electron configuration for each species.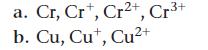
Transcribed Image Text:
Cr, Cr, Cr2+, Cr³+ a. b. Cu, Cut, Cu²+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a Ar4s3d ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Most of the second row transition metals do not follow the normal orbital filling pattern. Five of themNb, Mo, Ru, Rh, and Aghave a [Kr] 5s 1 4d x configuration and Pd has a [Kr] 4d 10 configuration....
-
When writing electron configurations, we use the building-up principle. However, there are exceptions to the building-up principle. An example is when a d-orbital will steal an electron from the...
-
Remaining time: 42:26 Question 1 What is the output of given code block? a = 15 b = 4 C = 3 print(a* b** c, c** 2, a % b - c, sep = "\\") Your answer: 960\\6\\0 960\6\15 960\9\15 180\9\0 960119110 O...
-
You are the senior auditor in charge of the December 31, 2018, year-end audit for Cleo Patrick Cosmetics Inc. (CPCI). CPCI is a large, privately held Canadian company that was founded in 1999 by one...
-
Nokia (www.nokia.com), the Finnish telecommunications company, has formed an equally owned joint venture with capital corporation, a state-owned Chinese company, to develop a center for the...
-
Describe the lean Six Sigma approach.
-
SWIGART v. BRUNO CALIFORNIA COURT OF APPEALS 13 CAL. APP. 5TH 529 2017 According to the American Endurance Ride Conference, endurance riding is a highly competitive and demanding sport. It is...
-
The Village of Harris issued $5,000,000 in 6 percent general obligation, taxsupported bonds on July 1, 2011, at 101. A fiscal agent is not used. Resources for principal and interest payments are to...
-
Prepare journal entry for December 31 adjusting entry. (Credit account titles are automatically indented when the amount is entered. Do not indent manually. If no entry is required, select "No Entry"...
-
Draw the Lewis diagrams for each ligand. Indicate the lone pair(s) that may be donated to the metal. Indicate any you expect to be bidentate or polydentate. a. CN b. Bipyridine (bipy), which has the...
-
Carbon monoxide and the cyanide ion are both toxic because they bind more strongly than oxygen to the iron in hemoglobin (Hb). Calculate the equilibrium constant value for this reaction: Does the...
-
Figure shows in cross section a machine having a rotor winding f and two identical stator windings a and b whose axes are in quadrature. The self-inductance of each stator winding is L aa and of the...
-
What do think about the ways in which most of us purchase and consume music in 2022 (streaming, downloading, finding new music via TikTok, etc.)? What about some of the "advantages" to consumers,...
-
Jing Company was started on January 1, Year 1 when it issued common stock for $38,000 cash. Also, on January 1, Year 1 the company purchased office equipment that cost $16,200 cash. The equipment was...
-
Bank XYZ has had the following gross profit (loss) results from its only three business lines for the past three years (amounts in $millions): Business Line Trading & Sales Payment & Settlement...
-
A firm's stock has a market capitalization of 120 million and an equity beta of 0.75. Bonds issued by the firm have a yield rate of 7.7%. Bonds with a similar debt rating has a default rate of 2.7%...
-
Using a margin account, 691 shares are short sold for $21 per share. The initial margin requirement is 28%. If the price of the stock falls to $17 per share, what is the margin in the account now?...
-
Sample Company is an aerospace manufacturer located in summer side, PEI. The company's income statements and balance sheets for the fiscal years ended August 31, 2003 and 2004 are attached. 1) In...
-
Compare and contrast licensing and subcontracting.
-
Two capacitors contain equal amounts of energy, yet one has twice the capacitance. How do their voltages compare?
-
Is a force needed to hold the plates of a charged capacitor in place? Explain.
-
Does the capacitance describe the maximum amount of charge a capacitor can hold, in the same way that a buckets capacity describes the maximum amount of water it can hold? Explain.
-
What will be the relationship between the coupon rate, the yield rate, and the yield to maturity rate for bonds sold below par? Use the coupon rate of 8% semiannual and the yield to maturity rate of...
-
They are ingenious solutions for those pressed to build a large number of related (but separate) instructional materials in short periods of time. a. Learning theories b. Learning strategies c....
-
1. You plan to buy a car. The price of the car is $85,700. You will make a down payment of $13,598. The remainder of the car's cost will be financed over a period of 5 years. You will repay the loan...

Study smarter with the SolutionInn App


