Use data from Appendix IIB to calculate S rxn for each of the reactions. In each case,
Question:
Use data from Appendix IIB to calculate ΔS°rxn for each of the reactions. In each case, try to rationalize the sign of ΔS°rxn.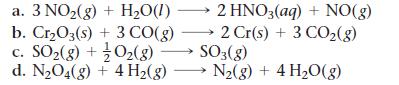
Appendix IIB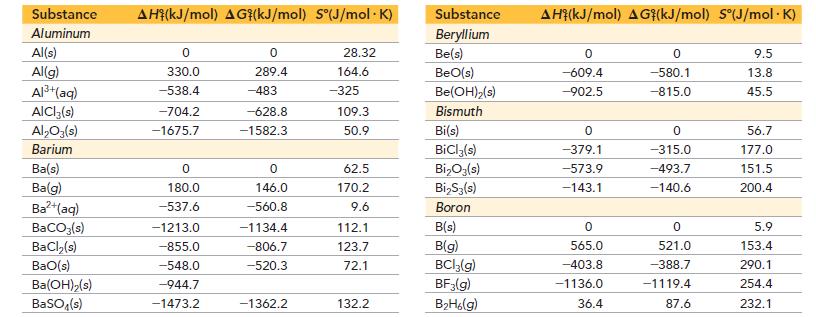
Transcribed Image Text:
a. 3 NO₂(g) + H₂O(1)→ b. Cr₂O3(s) + 3 CO(g) c. SO₂(g) + O₂(g) 2 HNO3(aq) + NO(g) 2 Cr(s) + 3 CO₂(g) SO3(g) d. N₂O4(g) + 4H₂(g) →→→ N₂(g) + 4H₂O(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a 3NO2g H2Ol 2HNO3aq NOg ASPx 2 SHNO3aq 1 SOg 3 SNOg 1 SO1 Substitute the values ...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use data from Appendix IIB to calculate S rxn for each of the reactions. In each case, try to rationalize the sign of S rxn . Appendix IIB a. CH4(g) + H(g) CH6(g) b. C(s) + HO(g) CO(g) + H(g) c....
-
Use data from Appendix IIB to calculate the equilibrium constants at 25 C for each reaction. G f for BrCl(g) is -1.0 kJ/mol. Appendix IIB a. 2 NO(g) NO4(8) b. Br(g) + Cl(g) = 2 BrCI(g)
-
Use data from Appendix IIB to calculate the equilibrium constants at 25 C for each reaction. Appendix IIB a. 2 CO(g) + O(g) = 2 CO(g) b. 2 HS(g) = 2 H(g) + S(g)
-
Q1-Mutual funds provide the following for their shareholders. A. diversification B. professional management C. record keeping and administration D. all of these options
-
Discuss the strategic implications of marketing in Mexico.
-
Given the accounting equation, answer each of the following questions. 1. The liabilities of Franks Smoothies are $120,000, and the stockholders equity is $325,000. What is the amount of Franks total...
-
Sigma Security, Inc., began the year with $15,000 of common stock and $32,000 of retained earnings. On May 5, investors bought $12,000 of additional stock in the business. On August 22, the business...
-
The demand curve for product X is given by Qdx = 460 = 4Px. a. Find the inverse demand curve. b. How much consumer surplus do consumers receive when Px = $35? c. How much consumer surplus do...
-
During 2021, Grouper Corporation spent $152.640 in research and development costs. As a result, a new product called the New Age Piano was patented. The patent was obtained on October 1, 2021, and...
-
Find S for the formation of CH 2 Cl 2 ( g) from its gaseous elements in their standard states. Rationalize the sign of S.
-
Rank each set of substances in order of increasing standard molar entropy (S). Explain your reasoning. a. 1(g); F(g); Br2(g); Cl(g) b. HO(g); HO(g); HS(g) c. C(s, graphite); C(s, diamond); C(s,...
-
Evaluate each of the following processes applied to cyclononatetraene, and decide whether the species formed is aromatic or not: (a) Addition of one more electron, to give C9H10 (b) Addition of two...
-
In the objective rational approach, only managers are qualified to think about strategy. Thus, strategic planning is at the top of the hierarchy ? Explain
-
Use MATLAB to calculate the following values. 1. Write a program to calculate the value. 1+2+3+4+...+1000 2. Write a program to calculate the values. 1+4+9+...+10000 3. Write a program to calculate...
-
Morrow Enterprises Inc. manufactures bathroom fixtures. Morrow Enterprises' stockholders' equity accounts, with balances on January 1, 20Y6, are as follows: Common stock, $20 stated value (500,000...
-
Answer questions 25,26,27 & 28 and show all work 24. The once-a-year order for each year's calendar arrives in September. From past experience the September-to-July demand for the calendars can be...
-
Analyze and discuss the estimated cost per cookie using job order costing, the estimated cost per cookie using process costing, and the estimated sales price per cookie. (for a batch of 500 sugar...
-
On March 1, 2017, Westmorlan Company acquired real estate on which it planned to construct a small office building. The company paid $75,000 in cash. An old warehouse on the property was razed at a...
-
Why is it necessary to study the diffusion of molecules in biological systems?
-
Consider two cylinders with the same density and the same length. If the ratio of their diameters is 1.5/1, what is the ratio of their moments of inertia?
-
In our examples with CDs (such as Example 8.7), we usually ignored the hole in the center. Calculate the moment of inertia of a CD, including the effect of the hole. For a CD of radius 6.0 cm,...
-
The moment of inertia for a square plate of mass M and length L that rotates about an axis perpendicular to the plane of the plate and passing through its center is ML 2 /6 (Fig. P8.49A). What is the...
-
The goals of the YCJA is to prevent crime, rehabilitate and reintegrate offenders, and to ensure meaningful consequences for actions. "To what extent does the YCJA meet the needs of Canadians? If you...
-
A 16-year-old girl by the name of Jenny Foster was found hitch-hiking along the Adirondack Mountains Highway in upstate New York. Mr. David Sykes, a driver on the highway, stopped to pick up the...
-
In the context of absorptive separation processes, how does the introduction of reactive absorption (e.g., using chemical reactants) alter the absorption equilibrium and kinetics? What are some...

Study smarter with the SolutionInn App


