Use data from Appendix IIB to calculate the equilibrium constants at 25 C for each reaction. Appendix
Question:
Use data from Appendix IIB to calculate the equilibrium constants at 25 °C for each reaction.![]()
Appendix IIB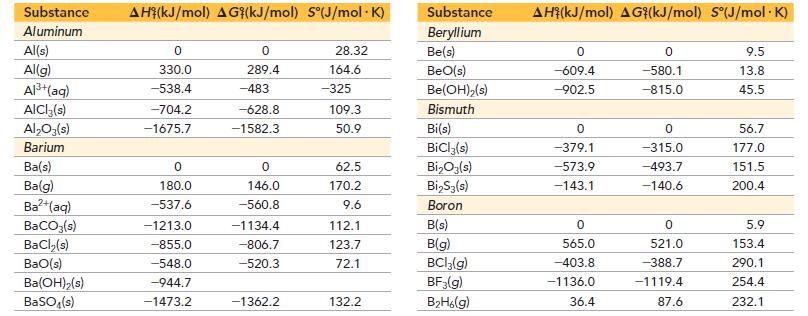
Transcribed Image Text:
a. 2 CO(g) + O₂(g) = 2 CO₂(g) b. 2 H₂S(g) = 2 H₂(g) + S₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a 148...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use data from Appendix IIB to calculate the equilibrium constants at 25 C for each reaction. G f for BrCl(g) is -1.0 kJ/mol. Appendix IIB a. 2 NO(g) NO4(8) b. Br(g) + Cl(g) = 2 BrCI(g)
-
Estimate the value of the equilibrium constant at 655 K for each reaction in Problem 74. (H f for BrCl is 14.6 kJ/mol.) Problem 74 Use data from Appendix IIB to calculate the equilibrium constants at...
-
Use data from Appendix C to calculate the equilibrium constant, K, at 298 K for each of the following reactions: H2(g) + 12(g) 2 HI(g) C2H5OH (g)- C2H4(g) + H2O(g)
-
8. A 12-tur square wire loop of 1-m length carrying a current of 1-A in the counter clockwise direction as shown in the figure. A magnetic field of magnitude 3000 G is directed into the board. Find,...
-
For each of the following transactions, state which account (s) would be debited and credited in a job order costing system for a desert landscaping business: 1. Charged customer for landscape design...
-
SuperQuick Computer Corporation reported the following income statement for a recent quarter. Consolidated Statement of Income For the Quarter Ended December 31, 2004 Sales ..................$719,150...
-
Why is it advisable to obtain a written confession from the subject of an investigation?
-
The debits to Work in ProcessRoasting Department for Morning Brew Coffee Company for August 2014, together with information concerning production, are as follows: All direct materials are placed in...
-
What is operating leverage and how is it related to the cost structure of any organisation ?
-
Which process results in the increase in entropy of the universe? a) The cooling of a hot cup of coffee in room temperature air b) The evaporation of water from a desk at room temperature c) The...
-
Without doing any calculations, determine the signs of S sys and S sur r for each chemical reaction. In addition, predict under what temperatures (all temperatures, low temperatures, or high...
-
Should efforts be made to formulate a model state code of environmental regulations?
-
A diver has 1.1 L of air in her lungs at an air temperature of 27.6C and a pressure of 1.02 atm. What is the volume (in L) of air in her lungs after she dives, while holding her breath, to a depth of...
-
Question 2 (23 marks) Calgary Drama Club (CDC) is a drama club for young Calgarians. It provides an opportunity for young people to learn about drama and perform in the theatre at levels consistent...
-
Where are manufactures and supplier's current product specifications and procedures usually located? Explain
-
lim f(1)-f(x) What is x1 x-1 if f(x) = ex? (1 point)
-
It is found that a saturated solution of sodium chloride in water boils under atmospheric pressure at 109C. Under an absolute pressure of 25.4 kPa, water boils at 65.6C and saturated sodium chloride...
-
During a coffee-room debate among several young MBAs who had recently graduated, one of the young executives flatly stated, The most this company can lose on its Brazilian division is the amount it...
-
Apply Jacobis method to the given system. Take the zero vector as the initial approximation and work with four-significant-digit accuracy until two successive iterates agree within 0.001 in each...
-
A solid sphere and a solid cylinder, both of mass m and radius R, are rolling without slipping with speed v. Find (a) The ratio of the angular momentum of the sphere to that of the cylinder and (b)...
-
An object is rolling without slipping, with a transnational kinetic energy of KE trans and a rotational kinetic energy of KE rot . If KE trans / KE rot = 3/2, what might this object be, (a) a hoop,...
-
A figure skater (Fig. 9.11) has an initial angular velocity of 3.8 rad/s. She then pulls her arms in, and her angular velocity increases to 9.5 rad/s. What is the ratio of her final kinetic energy to...
-
Suppose that Zamboni Enterprises is the only company that sells zambonis (ice resurfacing machines). To produce the machines, the company hires assembly workers. Since these workers can work in many...
-
Mckinsey Consultants have been advocating for businesses to adopt the Business Process Re-Engineering (BPR) methodology to influence their operational performance. Critically evaluate this...
-
summarize the devotion reading and discuss how this relates to your own life. Why is living with virtue important? What are some of the main distractions in life that keep us from living a virtuous...

Study smarter with the SolutionInn App


