Use the data shown here to find the equilibrium constant (K c ) for the reaction A(g)
Question:
Use the data shown here to find the equilibrium constant (Kc) for the reaction A(g) ⇌ 2 B(g) + C(g).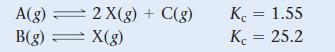

Transcribed Image Text:
A(g) = B(g) = 2X(g) + C(g) X(g) Kc = 1.55 Kc = 25.2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
d ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Determine A in the indicated figures. Fig. 2.40 (c) B 4 66 A (c) 4 C
-
The equilibrium constant Kc for the reaction equals 4.1 at 300oC. a. A sample of 35.8 g of PCl5 is placed in a 5.0-L reaction vessel and heated to 300oC. What are the equilibrium concentrations of...
-
The equilibrium constant Kc for the reaction is 3.8 Ã 10-5 at 727°C. Calculate Kc and KP for the equilibrium at the same temperature. 12(g)--21(g) 21(g)- 2(g)
-
Lead has one of the highest densities of all the pure metals. The density of lead is 11,340 kg/m. What is the density of lead in units of lbm/in?
-
Refer to Exercise 21-5. Assume the economic lot size for small casings is 30,000 and that of the large casings is 10,000. Wadley Manufacturing sells an average of 590 small casings per workday and an...
-
Sparrow Company had the following adjusted trial balance at December 31, 2009. Required: Prepare a single-step income statement for Sparrow Company for2009. Credit Accounts Receivable 40,000...
-
Following is a sample of five matched pairs. Let 1 and 2 represent the population means and let d = 1 2. A test will be made of the hypotheses H0: d = 0 versus H1: d > 0. a. Compute the differences....
-
Frio Company uses standard costs with its job order cost accounting system. In January, an order (Job No. 84) was received for 5,500 units of Product D. The standard cost of 1 unit of Product D is as...
-
Analyze Threats of New Entrants to the Market Objective: Perform a Porter's Five Forces analysis on the automotive industry. In order to make your recommendation about a new industry for diversifying...
-
Write an expression for the equilibrium constant (K c ) for this chemical equation: CaCO3(s) = CaO(s) + CO2(g)
-
Consider the reaction A(g) B(g). The images shown here illustrate equilibrium mixtures of A (red) and B (black) at three different temperatures. At which temperature is the equilibrium constant the...
-
What types of transportation challenges must organizations take into account when considering global sourcing?
-
Draw EERD-A database is required for judiciary to keep record of law. The organization wants to keep record of prisoner and their cases information. The prisoner has the unique number name address...
-
Assuming the students in your class are a representative sample of the US population, what is the likelihood you or a classmate is dealing with a high degree of communicative apprehension?
-
What are the key differences between aerobic and anaerobic respiration, particularly concerning the final electron acceptor and overall energy yield ? Explain
-
(a) Determine how the temperature changes during an isobaric process if the volume is tripled. (b) Determine how the volume changes during an isothermal process if the pressure is reduced by one...
-
Select one of the formats for analyzing, sorting, and interpreting data Research and discuss how the format is used in a business application. Provide an example in your discussion. Why do you think...
-
Following is the income statement of Jericho Co. for its most recent scal year. Sales........................................................ $267,000 Cost of Goods...
-
The liquidliquid extractor in Figure 8.1 operates at 100F and a nominal pressure of 15 psia. For the feed and solvent flows shown, determine the number of equilibrium stages to extract 99.5% of the...
-
In the circuit in Fig. 6.64 , let i s = 4.5e 2t mA and the voltage across each capacitor is equal to zero at t = 0. Determine v 1 and v 2 and the energy stored in each capacitor for all t > 0. 36 24...
-
If v(0) = 0, find v(t), i 1 (t), and i 2 (t) in the circuit of Fig. 6.63 . Is (mA). 30 5 t -30 6 F is 4F
-
Assuming that the capacitors are initially uncharged, find v o (t) in the circuit of Fig. 6.62 . (mA) A 6 F 90 is 3 F vo(t) 2 t(s)
-
Liz purchased two acres of lakefront property from her ex-husband, Richard, paying $500,000. Richard conveyed the property via a quitclaim deed. Liz's attorney dutifully recorded the deed the next...
-
Consider a dataset with 2 points in ID: (x = 0, y = -1) and (x2 = 2, y2 = 1). Map each point to 3D using the feature vector (x) = [1, 2x, x]. This is equivalent to using a second order polynomial...
-
Describe 2 change models that you could use to create change in an organization. Choose 1 of the models that you think would be most successful in an organization, and analyze reasons why you chose...

Study smarter with the SolutionInn App


