Use the vant Hoff factors in Table 14.9 to calculate each colligative property: a. The melting point
Question:
Use the van’t Hoff factors in Table 14.9 to calculate each colligative property:
a. The melting point of a 0.100 m iron(III) chloride solution
b. The osmotic pressure of a 0.085 M potassium sulfate solution at 298 K
c. The boiling point of a 1.22% by mass magnesium chloride solution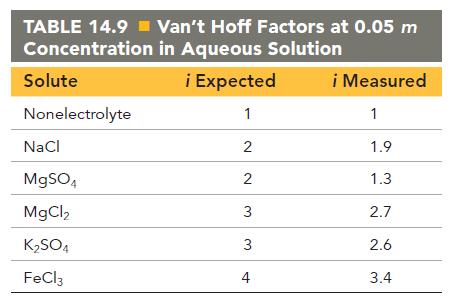
Transcribed Image Text:
TABLE 14.9 Van't Hoff Factors at 0.05 m Concentration in Aqueous Solution i Expected 1 Solute Nonelectrolyte NaCl MgSO4 MgCl, KSO4 FeCl3 2 2 3 3 4 i Measured 1 1.9 1.3 2.7 2.6 3.4
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a 063...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Using the vant Hoff factors in Table 14.9, calculate the mass of solute required to make each aqueous solution: a. A sodium chloride solution containing 1.50 * 10 2 g of water that has a melting...
-
A sample containing an alkali sulfate is dried, weighed and dissolved in dilute HCl. Barium chloride solution is added in excess to precipitate barium sulfate, and the precipitate is digested in the...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
How many orders of magnitude is 3 . 2 \ times 1 0 - 9 m smaller than 0 . 0 0 0 0 4 m ?
-
Jack Merritt is the controller for Universal Concrete Products (UCP), a manufacturing company with headquarters in Columbus, Ohio. UCP has seven concrete product plants located throughout the Midwest...
-
A cylindrical rod of copper originally 16.0 mm (0.625 in.) in diameter is to be cold worked by drawing; the circular cross section will be maintained during deformation. A cold-worked yield strength...
-
A company wishes to hedge its exposure to a new fuel whose price changes have a 0.6 correlation with gasoline futures price changes. The company will lose $1 million for each 1 cent increase in the...
-
Cannondale Company purchased an electric wax melter on April 30, 2014, by trading in its old gas model and paying the balance in cash. The following data relate to the purchase. List price of new...
-
A child bounces a 48 g superball on the side- walk. The velocity change of the superball is from 23 m/s downward to 13 m/s upward. If the contact time with the sidewalk is 1 800 s, what is the...
-
A 1.2 m aqueous solution of an ionic compound with the formula MX 2 has a boiling point of 101.4 C. Calculate the vant Hoff factor (i) for MX 2 at this concentration.
-
Determine the required concentration (in percent by mass) for an aqueous ethylene glycol (C 2 H 6 O 2 ) solution to have a boiling point of 104.0 C.
-
A model of a spillway is to be built at 1/80 scale. If the prototype has a discharge of 800 m 3 /s, what must be the water discharge in the model to ensure dynamic similarity? The total force on part...
-
General budgets are most common for which of the following funds? a. General Fund. b. Capital Projects Fund. c. Permanent Fund. d. Internal Service Fund.
-
In the Statement of Revenues, Expenditures, and Changes in Fund Balances, transfers must be reported a. in a separate section immediately following revenues. b. in a section immediately following the...
-
Appropriation requests for the General Fund are commonly approved, controlled, accounted for, and reported in each of the following expenditure categories except: a. Function or program. b....
-
The following GAAP requirements for budgetary reporting are true except a. budgetary comparisons for the General Fund and major Special Revenue Funds must include expenditure data that is at least as...
-
Which of the following statements would be true concerning budgetary integration? a. The integration of budgetary accounts into the general ledger does not affect the asset and liability accounts. b....
-
Esme Companys management is trying to decide whether to eliminate Department Z, which has produced low profits or losses for several years. The companys 2013 departmental income statement shows the...
-
What key concerns must functional tactics address in marketing? Finance? POM? Personnel?
-
Light having a vacuum wavelength of 600 nm, traveling in a glass (n g = 1.50) block, is incident at 45 on a glassair interface. It is then totally internally reflected. Determine the distance into...
-
Derive an expression for the speed of the evanescent wave in the case of internal reflection. Write it in terms of c, n i , and i .
-
A large block of diamond is covered, on top, by a layer of water. A narrow beam of light travels upward in the solid and strikes the solidliquid interface. Determine the minimum incident angle that...
-
answered. Problem14This problem is current being displayed. Problem15 Question Content AreaStephen makes and sells cakes through bakeries. He knows he must sell 450 cakes a month to break even. Every...
-
year 1: net income $,2377 stockholders equit ending balance $10843, year2: net income 2350 stockholders equit ending balance $12332, year3: net income $,2679 stockholders equit ending balance $11079,...
-
Upon the conclusion of a 100,000-square-foot office building project, Casa Loma Development sold the property to Blue Jays Group. Subsequent to this transaction, Casa Loma Development and Blue Jays...

Study smarter with the SolutionInn App


