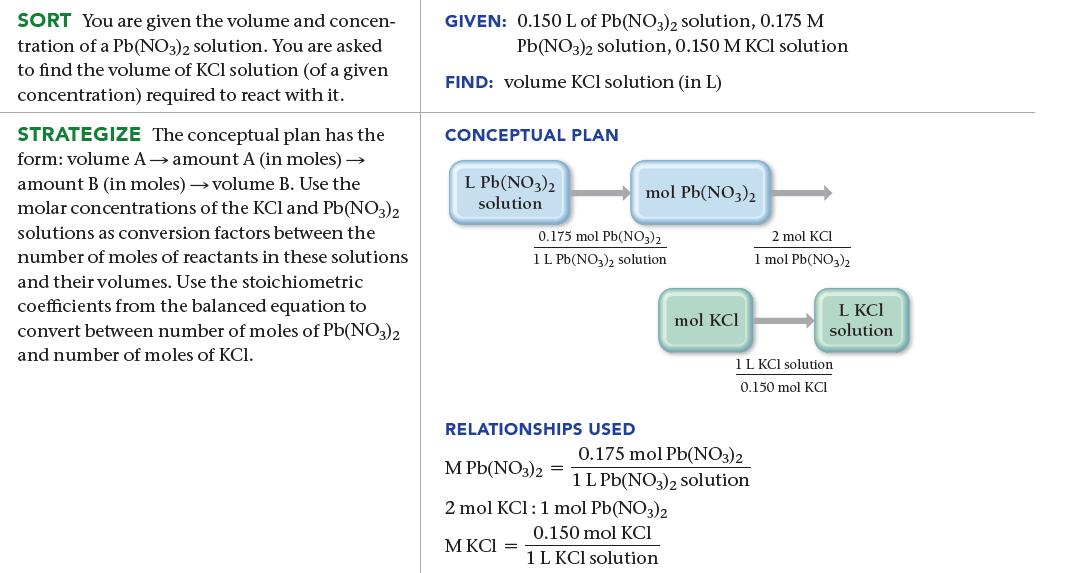
What volume (in L) of a 0.150 M KCl solution will completely react with 0.150 L of
Question:
What volume (in L) of a 0.150 M KCl solution will completely react with 0.150 L of a 0.175 M Pb(NO3)2 solution according to the following balanced chemical equation?
![]()
Transcribed Image Text:
2 KCl(aq) + Pb(NO3)2(aq) PbCl₂(s) + 2 KNO3(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
0150 L PbNO32 solution X 2 mol KCI 1 mol PbNO32 0175 mol PbNO32 1 LPbNO32 solution 1 L KCl solu...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
1. What mass of H2 should be produced by the reaction of Al with 75.0 mL of 2.95M HCl? 2Al(s) + 6HCl(aq) 2AlCl3(aq) + 3H2 (g). ln the lab, 0.15g H2 was collected. What is the % yield of the...
-
Twelve years ago, Birch Ltd. (BL) borrowed $480,000 from Oak Trust Inc. (OTI). The 12- year, 10% note is due on todays date, December 31, 2020. The note was originally issued at par. BL is unable to...
-
What volume of 0.512 M NaOH will react with 17.9 g of H2C2O4(s) according to the following chemical equation? H2C2O4(s) + 2NaOH(aq) ( Na2C2O4(aq) + 2H2O()
-
In the Young's double slit experiment, interference fringes are formed using Na-light of 589 nm and 589.5 nm. Obtain the region on the screen where the fringe pattern will disappear. You may assume...
-
It's Yours Manufacturing wishes to maintain a growth rate of 8 percent a year, a debt-equity ratio of 0.45, and a dividend payout ratio of 25 percent. The ratio of total assets to sales is constant...
-
Each person in one of your accounting classes is required to write a report on an accounting topic. Included in the report must be a discussion from a specific library book. When you go to the...
-
What are the two tests for determining whether a party may be joined in a lawsuit?
-
Margaret Cleary is a paralegal who works for a law firm that requires her to bill clients seven hours a day. One of Margarets assignments was to prepare interrogatories on a real estate case. She...
-
1. Identify one factor that influences how long a competitive advantage lasts. Provide one company example to explain why and how this factor influences the duration of the company's competitive...
-
The following unadjusted trial balance is for Ace Construction Co. as of the end of its 2013 fiscal year. The June 30, 2012, credit balance of the owners capital account was $53,660, and the owner...
-
Explain how a strong electrolyte, a weak electrolyte, and a nonelectrolyte differ.
-
The image shown at the far right represents a small ANSWER NOW! volume within 500 mL of aqueous ethanol (CH 3 CH 2 OH) solution. (The water molecules have been omitted for clarity.) Which of the...
-
Suppose we start with a Jeffreys' prior for the standard deviation . What value of S x an inverse chi-squared(k) will we use? (a) Find the posterior using the macro NVarICP in Minitab or the function...
-
Identify five users of audited financial statements. Briefly indicate why independ- ently audited financial statements are important to each user.
-
An audit committee should do all of the following except: a. Decide whether to retain or dismiss the outside auditors. b. Determine whether material fraud ought to be reported in the company's fi-...
-
Which of the following is not a Sarbanes-Oxley requirement for audit commit- tees? a. The audit committee must be chaired by the chair of the board of directors. b. Audit committee members must be...
-
Let the balance sheet of the bank be described by L + R + T = D + E, where L is the stock of loans, R is the reserves, T is the stock of liquid assets, D is the deposits and E is the equity capital....
-
Which of the following would not be a correct statement about a partial cause of corporate governance failures? a. Boards of directors approved stock option plans that did not align management and...
-
Leah's Toys also makes rubber balls. The current process is capable of producing balls that weigh, on average, 3 ounces, with a standard deviation of 0.25 ounces. What is the process capability...
-
Explain briefly what is meant by electronic data interchange (EDI). How does EDI affect a companys audit trail?
-
Predict the major product(s) for each of the following reactions: (a) (b) (c) (d) (e) (f) CH NH2 [H*] (-H20) 1) PhMgBr 2) H20
-
Identify the starting materials needed to make each of the following acetals: (a) (b) (c) OEt
-
Using ethanol as your only source of carbon atoms, design a synthesis for the following compound:
-
Define SPARCS and discuss the following: 1. Purpose - why was it developed. 2. Review data elements collected. 3. Identify how this data is meaningful to New York State and other healthcare...
-
Describe the complexities related to expatriate executive assignments and the complexity of the international compensation balance. Include in the analysis the perquisites management. Describe the...
-
How does Ford Motor Company epitomize the 6 elements of a learning organization? How might it incorporate any missing elements or improve some weaker elements to be a stronger learning organization?...

Study smarter with the SolutionInn App


