Identify the starting materials needed to make each of the following acetals: (a) (b) (c) OEt
Question:
(a)
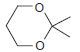
(b)
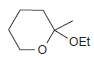
(c)
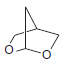
Transcribed Image Text:
OEt
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
a b...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Provide the starting materials needed to synthesize each compound by acylation of an enolate. (a) (b) (c) CO2Et
-
Identify the starting aldehyde or ketone needed to make each of the following compounds via an aldol condensation. (a) (b) (c)
-
Identify the starting alkene necessary to make each of the following amino acids using an asymmetric catalytic hydrogenation. (a) l-alanine (b) l-valine (c) l-leucine (d) l-tyrosine
-
Find each product. -3a+(4 + a)
-
Discuss the following topics in relation to brands: 1. What elements have contributed to the success of the brand? 2. What role did packaging play in the brands success? 3. How have the owners of...
-
What information should be requested by the auditors from the trustee responsible for an issue of debentures payable?
-
(a) Show that the Jones matrix of a polarization analyzer set at angle \(\alpha\) to the \(X\)-axis is given by \[ \underline{\mathbf{L}}(\alpha)=\left[\begin{array}{cc} \cos ^{2} \alpha & \sin...
-
Harrison Company makes two products and uses a traditional costing system in which a single plantwide predetermined overhead rate is computed based on direct labor-hours. Data for the two products...
-
Research Chipotle In a document what are chipotle internal strengths and internal weaknesses? What are some market opportunities and market threats? What is chipotle strategy? If you are introducing...
-
Why perform a retirement needs analysis?
-
Predict the major product(s) for each of the following reactions: (a) (b) (c) (d) (e) (f) CH NH2 [H*] (-H20) 1) PhMgBr 2) H20
-
Using ethanol as your only source of carbon atoms, design a synthesis for the following compound:
-
The balance sheet for Tactex Controls Inc., provincially incorporated in 2021, reported the following components of equity on December 31, 2022. Required 1. Prepare journal entries to record the...
-
What determines the order of the information shown on the balance sheet?
-
A retail clothing firm is evaluating the development of a new range of all-weather coats. These coats contain an internal solar battery to provide heating whenever the garment is worn. The solar...
-
What are the two ways a sinking fund can be handled? Which method will be chosen by the firm if interest rates have risen? If interest rates have fallen?
-
Indicate whether each of the following statements is true or false by writing T or F i n t he a nswer c olumn. A newspaper that prints a story about a public fi gure, with knowledge that the story is...
-
Define floating-rate bonds and zero coupon bonds.
-
Use synthetic division to divide (x) by x - k for the given value of k. Then express (x) in the form (x) = (x - k)q(x) + r. (x) = 2x 3 + 3x 2 - 16x + 10; k = -4
-
Chris Zulliger was a chef at the Plaza Restaurant in the Snowbird Ski Resort in Utah. The restaurant is located at the base of a mountain. As a chef for the Plaza, Zulliger was instructed by his...
-
Classify each of the following substance according to their solvent properties (as in Table 8.2.) (a) 2,2,2-triftuoroethanol ( - 26) (b) 2,2,4-trimethylpentane ( : 2)
-
The experiment is repeated, except that I mL of l-nonanol is added instead of methanol. In which layer is the alcohol dissolved? Explain.
-
A widely used undergraduate experiment is the recrystallization of acetanilide from water. Acetanilide (see following structure) is moderately soluble in hot water, but much less soluble in cold...
-
Recently, there has been a resurgence in labor union organization and a subsequent increase in union membership across the United States. What can today's labor unions learn from their past 140-year...
-
). Prove by contraposition: "Vm, ne Z, if m -n is even, then m - n is even (15 points)
-
The production function is Y = AKL-a (1) where L is labor at time t, K, is capital at time t, A, is TFP at time t and a (0, 1). The law of motion for capital is K = 8Y-8K Assume that TFP evolves...

Study smarter with the SolutionInn App


