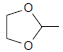
Using ethanol as your only source of carbon atoms, design a synthesis for the following compound:
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
HSO4 PCC ...View the full answer

Answered By

RISHABH YADAV
I have been tutoring since more than 2 years. I teach mathematics and physics. My students have qualified a number of examinations that have gained them jobs. Some of my friends regularly keep in touch with me so as to get their doubts cleared easily and with a friendly tone. My students have never complained of being late for classes or foe not considering their issues because I always keep my tutoring and my students at topmost priority.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Sciences questions
-
Using ethanol as your only source of carbon atoms, propose a synthesis for each of the following compounds. (a) (b) OEt .
-
Using toluene as your only source of carbon atoms, show how you would prepare the following compound.
-
Using acetylene as your only source of carbon atoms, design a synthesis of trans-5- decene:
-
Simplify each expression. Assume that all variables represent nonzero real numbers. (xy-3)-2
-
You are the Certified Managerial Accountant (CMA) for Northwood Company. You have worked for the company for several years and your responsibilities include working with managers to analyze business...
-
Can the client change a set of financial statements to receive an unmodified opinion instead of an opinion qualified as to the adequacy of disclosure? Explain.
-
Starting with Eq. (4.1-10), show that if \(\Delta v \ll \bar{v}\) and \(r \ll 2 c / \Delta v\) for all \(P_{1}\), then \[ \mathbf{u}\left(P_{0}, t ight) \approx \iint_{\Sigma} \frac{e^{j 2 \pi(r /...
-
Trish Craig and Ted Smith have a partnership and share income and losses in a 3:1 ratio. They decide to liquidate their partnership on December 31, 2014, when the balance sheet shows the following:...
-
Zeing Manufacturing pays taxes of 20% on all pre-tax income up to and including $200,000. For pre-tax income above $200,000, the tax rate is 32%. If Zeing Manufacturing's contribution margin ratio is...
-
Hill-O-Beans Coffee Company blends four component beans into three final blends of coffee: one is sold to luxury hotels, another to restaurants, and the third to supermarkets for store label brands....
-
Identify the starting materials needed to make each of the following acetals: (a) (b) (c) OEt
-
Propose an efficient synthesis for each of the following transformations: (a) (b)
-
1. Do the differenced series appear stationary according to the augmented Dickey{Fuller tests? 2. Do you see evidence of autocorrelations in the differenced series? If so, describe these correlations.
-
What four types of financial statements does the annual report typically include?
-
Indicate whether each of the following statements is true or false by writing T or F i n t he a nswer c olumn. Slander is a spoken, and libel is a written, form of defamation.
-
To what types of risk are investors of foreign bonds exposed?
-
Indicate whether each of the following statements is true or false by writing T or F i n t he a nswer c olumn. Offshore gambling refers to foreign citizens placing bets in casinos located within the...
-
What is the statement of stockholders equity, and what information does it provide?
-
Use synthetic division to divide (x) by x - k for the given value of k. Then express (x) in the form (x) = (x - k)q(x) + r. (x) = 2x 3 + x 2 + x - 8; k = -1
-
Four GWU students have been selected to taste food sold by 3 different food trucks labeled as food truck A, B and C on H & 22nd Streets every Monday for 3-weeks. For each student, food trucks are...
-
Give the structure of each of the following compounds. (a) Chlorocyclopropane (b) Methylene iodide
-
Acetone (Table 8.2) has a significant dipole moment (2.7 D). Using structures, show the stabilizing interactions to be expected between acetone solvent molecules and (a) A dissolved potassium ion;...
-
Give the structure of A lecithin.
-
What advantage does the private resident buying oce (store-owned offices) aord its stores that independent buying oces do not? Describe two functions of an independent buying office. Describe the 4...
-
10. Consider the following data - == - -5 1 3 4 -1 0 1 1 (a) Find the Least Squares Regression Line. (b) Find the coefficient of correlation and determination. (c) Find a 90% confidence interval for...
-
Which point on the graph y=4x are closest to the point (0, 2)? (Hint: consider the distance formula between two points)

Study smarter with the SolutionInn App


