Fig. 15.3 outlines the classic scheme for separating a mixture of insoluble chloride salts from one another.
Question:
Fig. 15.3 outlines the classic scheme for separating a mixture of insoluble chloride salts from one another. Explain the chemistry involved in the various steps of the figure.
Fig. 15.3
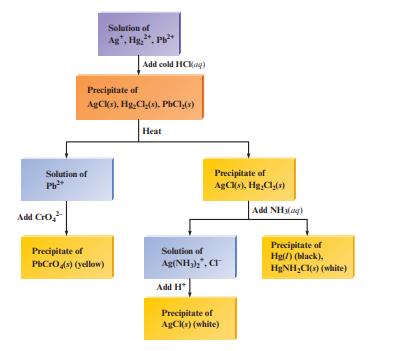
Transcribed Image Text:
Solution of Pb²+ Add CrO₂²- Solution of Ag, Hg., Pb²+ Precipitate of AgCl(s), Hg,Cl(s), PbCl(s) Precipitate of PbCrO4(s) (yellow) Add cold HCKag) Heat Precipitate of AgCl(s), Hg₂Cl(s) Solution of Ag(NH3)2, CT Add H Precipitate of AgCl(s) (white) Add NH3(g) Precipitate of Hg(/) (black). H₂NH₂Cl(s) (white)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The figure you sent me appears to show a separation scheme for a mixture of three ca...View the full answer

Answered By

Rukhsar Ansari
I am professional Chartered accountant and hold Master degree in commerce. Number crunching is my favorite thing. I have teaching experience of various subjects both online and offline. I am online tutor on various online platform.
5.00+
4+ Reviews
17+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Fig. 15.2 summarizes the classic method for separating a mixture of common cations by selective precipitation. Explain the chemistry involved with each of the four steps in the diagram. Fig. 15.2...
-
Outline a procedure for separating a mixture of benzoic acid, 4-methylphenol, aniline,and benzene using acids, bases, and organic solvents.
-
Devise a procedure for separating a mixture of the four stereoisomers of isoleucine into its four components: (+)-isoleucine, (-)-isoleucine, (+)-alloisoleucine, and (-2)-alloisoleucine (Problem 31)....
-
Each of the systems in Problems 11 through 18 has a single critical point (x 0 , y 0 ). Apply Theorem 2 to classify this critical point as to type and stability. Verify your conclusion by using a...
-
If the claimants had refuted the governments assertion that the cash was illicit, would the result in this case have been different? Why or why not?
-
The tar content in 30 samples of cigar tobacco follows: (a) Is there evidence to support the assumption that the tar content is normally distributed? (b) Find a 99% CI on the mean tar content. (c)...
-
Maribel Baltazar was hired by clothing retail merchandiser Forever 21 in 2007. During the hiring process, Baltazar was given an 11-page document to sign, two pages of which contained an arbitration...
-
Classify each of the following items of factory overhead as either a fixed or a variable cost. (Include any costs that you consider to be semivariable within the variable category. Remember that...
-
Explain how the Pakistan crisis will affect the following companies: a) MEIR Commodities India Pvt. Ltd., an agri commodity trading house exporting sugar, molasses, organics, etc. b) Honda, Toyota,...
-
Go through the transaction list (Document1) and identify which cycle each transaction belongs to. A transaction will belong to only one cycle; either the Sale to Cash Receipt (S/CR) Cycle, Purchases...
-
Calculate the molar solubility of Cd(OH) 2 , K sp = 5.9 10 -11 .
-
Under what circumstances can you compare the relative solubilities of two salts directly by comparing the values of their solubility products? When can relative solubilities not be compared based on...
-
Listed are the sales at Robertas Ice Cream Stand for the last 5 years, 2015 through 2019. Year Sales 2015 . . . . . . . . . . . . . . . $130,000 2016 . . . . . . . . . . . . . . . 145,000 2017 . . ....
-
The density-speed relationship for a freeway lane was found to be: u 54.5-0.24k Speed is given by u, density is given by k, and flow is given by q. Determine: a. speed at capacity b. density at...
-
State your ideas/thoughts using one to two sentences only. 1.How can we fill our family life with love? 2.Everyone basically wants to have a stable family, peaceful family life. But in marriage and...
-
Can you please explain in simple words what is to do in this assignment.. like how should we present the leadership strategies? I only want to make sure I understand the assignment. The students...
-
You sell a forward on gold with a forward price F(0)=$1750. Spot gold is traded at S(0)=$1725. Gold price at expiration is S(T)=$1800 at expiration. Your payoff is (you receive) ...
-
How is Covid impacting or impacted the aviation industry? 1. what kind of a material response was needed to address the situation. 2. what kind of a communication-based response was needed to address...
-
Shilstone Supply, Inc. manufactures a variety of pumps and valves that are distributed through several thousand plumbing supply houses, as well as 100 manufacturer's representatives. Due to lessthan...
-
Reichenbach Co., organized in 2018, has set up a single account for all intangible assets. The following summary discloses the debit entries that have been recorded during 2018 and 2019. Instructions...
-
Draw a mechanism for the following transformation: NaOH, heat
-
The reaction in the previous problem is an equilibrium process. Draw a mechanism of the reverse process. That is, draw a mechanism showing conversion of the conjugated, cyclic enone into the acyclic...
-
When 2,6-heptanedione is heated in the presence of aqueous sodium hydroxide, a condensation product with a six-membered ring is obtained. Draw the product and show a mechanism for its formation.
-
A 10-foot U-Haul truck for in-town use rents for $14.95 per day plus $1.99 per mile. You are planning to rent the truck for just one day. a. Write the total cost of rental as a linear function of the...
-
1. To be a pilot in the United States Air Force, a candidate must meet a battery of physical requirements. One requirement is the candidate must have a standing height between 65 and 77 inches,...
-
1) How many milligrams would a 118 pound and 12-ounceRottweiler-Pit mix weigh? 2) Lets say the dog ate some cat poop in the yard and now needsan antiparasitic prophylactic. The solution strength on...

Study smarter with the SolutionInn App


