Without using Fig. 3.4, predict the order of increasing electronegativity in each of the following groups of
Question:
Without using Fig. 3.4, predict the order of increasing electronegativity in each of the following groups of elements.
Data in Fig. 3.4
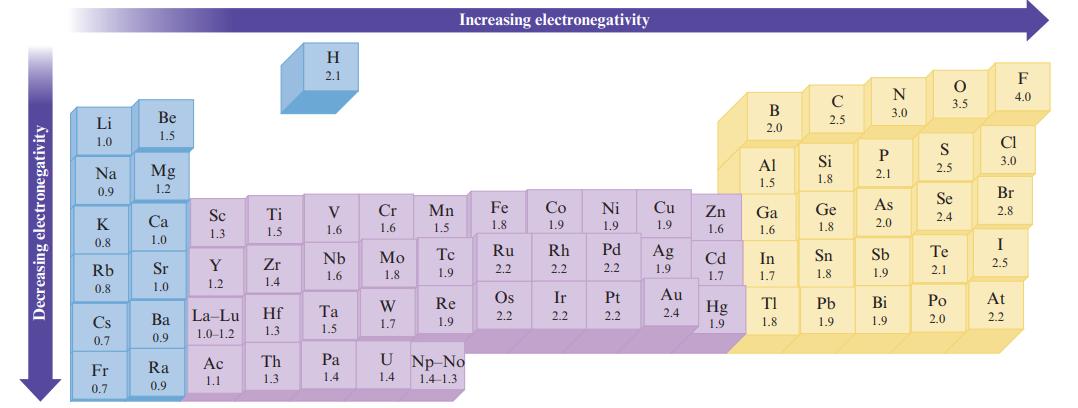
a. Na, K, Rb
b. B, O, Ga
c. F, Cl, Br
d. S, O, F
Transcribed Image Text:
Decreasing electronegativity Li 1.0 Na 8 0.9 K 0.8 Rb 0.8 Cs 0.7 Fr 0.7 Be 1.5 Mg 1.2 Ca 1.0 Sr 1.0 Ba 0.9 Ra 0.9 Sc 1.3 Y 1.2 La-Lu 1.0-1.2 Ac 1.1 Ti 1.5 Zr 1.4 Hf 1.3 Th 1.3 H 2.1 V 1.6 Nb 1.6 Ta 1.5 Pa 1.4 Cr 1.6 Mo 1.8 W 1.7 Mn 1.5 Tc 1.9 Re 1.9 Increasing electronegativity U Np No 1.4 1.4-1.3 Fe 1.8 Ru 2.2 Os 2.2 Co 1.9 Rh 2.2 Ir 2.2 Ni 1.9 Pd 2.2 Pt 2.2 Cu 1.9 Ag 1.9 Au 2.4 Zn 1.6 Cd 1.7 Hg 1.9 B 2.0 Al 1.5 Ga 1.6 In 1.7 ΤΙ 1.8 C 2.5 Si 1.8 Ge 1.8 Sn 1.8 Pb 1.9 P 2.1 N 3.0 As 2.0 Sb 1.9 Bi 1.9 S 2.5 O 3.5 Se 2.4 Te 2.1 Po 2.0 Cl 3.0 F 4.0 Br 2.8 I 2.5 At 2.2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
Without looking at Fig 34 Id predict the order of increas...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Without using Fig. 13.3, predict the order of increasing electronegativity in each of the following groups of elements. a. C, N, O b. S, Se, Cl c. Si, Ge, Sn d. Tl, S, Ge e. Na, K, Rb f. B, O, Ga...
-
Without using Fig. 3.4, predict which bond in each of the following groups will be the most polar. Fig. 3.4 a. C-H, Si-H, Sn-H b. Al-Br, Ga-Br, In-Br, Tl-Br c. C-O or Si-O d. O-F or O-CI Decreasing...
-
Without using Fig. 3.4, predict which bond in each of the fol- lowing groups will be the most polar. Data in Fig. 3.4 a. C-F, Si-F, Ge-F b. P-Cl or S-Cl c. S-F, S-Cl, S-Br d. Ti-Cl, Si-Cl, Ge-Cl...
-
Capital Inc. has prepared the operating budget for the first quarter of 2015. They forecast sales of $50,000 in January, $60,000 in February, and $70,000 in March. Variable and fixed expenses are as...
-
Why does a monopolist not charge the same price for the same good in two different countries?
-
Water flows in a 5-m-wide channel with a speed of \(2 \mathrm{~m} / \mathrm{s}\) and a depth of \(1 \mathrm{~m}\). The channel bottom slopes at a rate of \(1 \mathrm{~m}\) per \(1000 \mathrm{~m}\)....
-
True or False. The Euler-Bernoulli beam theory is more accurate than the Timoshenko theory.
-
Eastside Aquarium, Inc., manufactures and sells aquariums, water pumps, and air filters. The sales mix is 1:2:2 (i.e., for every one aquarium sold, two water pumps and two air filters are sold)....
-
Suppose that we have two machines A and B. The following table shows the execution times for programs that make up a benchmark suite for machine A, machine B, and a reference machine X. Program Exec...
-
Describe the type of bonding that exists in the Cl 2 (g) molecule. How does this type of bonding differ from that found in the HCl(g) molecule? How is it similar?
-
On March 7, 2007, Toby Young was participating in a five-day cross-country ski and yoga program with Elderhostel and the Craftsbury Center, Inc. Young spent the afternoon cross-country skiing. A...
-
Lego Group in Bellund, Denmark manufactures Lego toy construction blocks. The company is considering two methods for producing special purpose Lego parts. Method 1 will have an initial cost of...
-
A sound wave travels through a tube from a source to a detector. As shown in the figure, the wave breaks into two parts at point A: one part travels through the half-circle, the other through the...
-
Universal Maintenance Co. issues $700,000 of 9.50% bonds on July 1, 2020. Additional information on the bond issue is as follows: Bond date January 1, 2020 Maturity date January 1, 2030 Yield rate...
-
In many ways, we continue to discover how plants are similar to human. One of the interesting similarities includes the presence of consciousness in both humans and plants. With the help of examples,...
-
Noise-cancelling headphones are a kind of headphones which decrease the amount of background noise that get into your ears when you wear them. On what physical phenomena do these headphones depend?...
-
4. An air column which is closed at one end resonates at the third resonant length. The frequency of sound wave is 1244 Hz. The air temperature is 16C. a) What is the speed of sound in air? [APP - 2...
-
Jan Merkle is a fraud investigator who was working alone for the first time on an assignment. In previous cases, she had always worked under one of the senior partners in her forensic accounting...
-
1. Using the information from Problem 16-4B, prepare a statement of cash flows for Lim Garden Supplies Inc. using the direct method of presenting cash flows from operating activities. 2. How does Lim...
-
When Na 3 PO 4 (aq) is added to a solution containing a metal ion and a precipitate forms, the precipitate generally could be one of two possibilities. What are the two possibilities?
-
Sodium chloride is listed in the solubility rules as a soluble compound. Therefore, the K sp value for NaCl is infinite. Is this statement true or false? Explain.
-
When 5 M ammonia is added to a solution containing Cu(OH) 2 (s), the precipitate will eventually dissolve in solution. Why? If 5 M HNO 3 is then added, the Cu(OH) 2 precipitate re-forms. Why? In...
-
Core Co. leased a piece of manufacturing equipment from E-So Co. with the following terms: Annual lease payment: $770,000 Term of lease: 5 years Interest rate: 4% Lease commences on January 1, 2023...
-
Arndt, Incorporated reported the following for 2024 and 2025 ($ in millions): Revenues Expenses 2024 $ 948 804. 2025 $ 1,040 860 Pretax accounting income (income statement) Taxable income (tax...
-
Bonita Company combines its operating expenses for budget purposes in a selling and administrative expense budget. For the first 6 months of 2022, the following data are available. 1. Sales: 23,200...

Study smarter with the SolutionInn App


