Without using Fig. 3.4, predict which bond in each of the fol- lowing groups will be the
Question:
Without using Fig. 3.4, predict which bond in each of the fol- lowing groups will be the most polar.
Data in Fig. 3.4
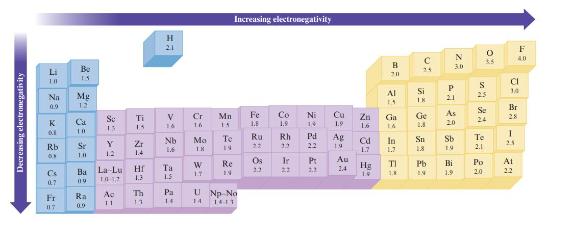
a. C-F, Si-F, Ge-F
b. P-Cl or S-Cl
c. S-F, S-Cl, S-Br
d. Ti-Cl, Si-Cl, Ge-Cl
Transcribed Image Text:
Decreasing electronegativity Li 18 K Na Mg 0.9 12 X 0. Cs 07 Be 20 Fr 117 Rb Se Y 1.2 de ²2:29 Ca 10 1.0 Ba 019 Ra Sc 13 0.9 Ti 15 Ac 11 Zr 1.4 La-Lut 10:12 1.3 = Th 14 H 21 V Nb 23 21 4: Hf Ta Pa 14 Cr 16 Mo IX W e p 1.7. Mn 14 To 19 Increasing electronegativity Re IN U Np No 14 1411 Fe 1.8 Ru 2# 8A 22 Os Co Ni 1.9 2:23 E Ir 22 14 Rh Pd 33 Pt 2.3 Cu 19 Ag 19 Au 24 398 25 1.6 Cd 1.7 B Zn Ga 14 Hg F 201 28 F3 TI с Si IK 33 1.8 Pb 30 27 29 82 N 21 As Sn Sb 18 20 19 Bi 1.9 0 S 25 Te 2.1 35 Se 24 Po 20 Cl 34 F 40 Br 28 -3 25. At 22
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
To predict which bond in each of the following groups will be the most polar we can use the followin...View the full answer

Answered By

Elias Gichuru
am devoted to my work and dedicated in helping my clients accomplish their goals and objectives,providing the best for all tasks assigned to me as a freelancer,providing high quality work that yields high scores.promise to serve them earnestly and help them achieve their goals.i have the needed expertise,knowledge and experience to handle their tasks.
4.80+
325+ Reviews
859+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Without using Fig. 3.4, predict which bond in each of the following groups will be the most polar. Fig. 3.4 a. C-H, Si-H, Sn-H b. Al-Br, Ga-Br, In-Br, Tl-Br c. C-O or Si-O d. O-F or O-CI Decreasing...
-
Without using Fig. 13.3, predict which bond in each of the following groups is the most polar. a. COF, SiOF, GeOF b. POCl, SOCl c. SOF, SOCl, SOBr d. TiOCl, SiOCl, GeOCl e. COH, SiOH, SnOH f. AlOBr,...
-
Use the electro negatively table (Figure) to predict which bond in each of the following sets is more polar, and indicate the direction of bond polarity for each compound. (a) H3C ? C1 OR C1 ? C1 (b)...
-
The following data applies to the two unrelated companies Lloyd Ltd and Cole Ltd: All taxable and deductible temporary differences relate to the profit or loss. Assume a corporate tax rate of 30%. A....
-
Given that real exchange rates fluctuate, when would be the best time to enter the market of a foreign country as an exporter to that market?
-
Determine values for the constants n and k (Equation 10.17) for the recrystallization of copper (Figure 10.11) at 102?C. y = 1 - exp(-kt") (10.17) 100 80 113C 102 C 88C 135C/ 119C 43C 60 40 20 102...
-
Refer to the data for Midway Cycles in E5-19A. Requirements 1. Compute the cost of goods sold under FIFO. 2. Compute the cost of goods sold under LIFO. 3. Which method results in the higher cost of...
-
Mom and Pop Groceries has just dispatched a years supply of groceries to the government of the Central Antarctic Republic. Payment of $250,000 will be made one year hence after the shipment arrives...
-
The National Consumer Commission (NCC) is currently investigating 'Lolli World & Co', the supplier of lollipops. The investigation commenced subsequent to the NCC receiving numerous complaints from...
-
Which of the following incorrectly shows the bond polarity? Show the correct bond polarity for those that are incorrect.
-
Describe the type of bonding that exists in the Cl 2 (g) molecule. How does this type of bonding differ from that found in the HCl(g) molecule? How is it similar?
-
If you see a house valued at $300,000 in Denver, find the price of a comparable house in Miami and Cheyenne. City Index City Index Denver Miami Phoenix Atlanta Baltimore 100 194 86 90 150 Boston Las...
-
a. What is meant by tick size? b. What is the tick size for common stock?
-
Explain expenditure-switching as a means to correct a current account deficit.
-
a. What is meant by "soft dollars"? b. What is the concern with soft dollars?
-
Make a list of the goods you commonly use that are imported. These can include food, clothing, electronics etc. If possible, identify the source of the goods.
-
Explain why a current account deficit may result in a depreciation of a country's currency.
-
Consider the sample of body temperatures (°F) listed in the last column of Data Set 3 in Appendix B. The summary statistics are given in the accompanying table. Use a 0.01 significance level to...
-
Rowland Textile Inc. manufactures two products: sweatshirts and T-shirts. The manufacturing process involves two activities: cutting and sewing. Expected overhead costs and cost drivers are as...
-
If a solution contains either Pb 2+ (aq) or Ag + (aq), how can temperature be manipulated to help identify the ion in solution?
-
Silver chloride dissolves readily in 2 M NH 3 but is quite insoluble in 2 M NH 4 NO 3 . Explain.
-
The stepwise formation constants for a complex ion usually have values much greater than 1. What is the significance of this?
-
what manner does the interaction between resilience and vulnerability dynamics contribute to nuanced understandings of adaptation within evolutionary frameworks ?
-
Fabric Plant: Budgeted overhead: $1,200,000 (50% fixed) Practical volume (direct labor hours): 120,000 hours Actual overhead: $1,150,000 (50% fixed) Actual hours worked (for each fabric type,...
-
Provide the missing data in the following table for a distributor of martial arts products: (Round "Turnover" and "ROI" answers to 1 decimal place.) Alpha Division Bravo Division Charlie Division...

Study smarter with the SolutionInn App


