Answer the following questions based on Figure 6.2: (a) Which type of radiation involves less energy, x-rays
Question:
Answer the following questions based on Figure 6.2:
(a) Which type of radiation involves less energy, x-rays or microwaves?
(b) Which radiation has the higher frequency, radar or red light?
(c) Which radiation has the longer wavelength, ultraviolet or infrared light?
Data given in figure 6.2
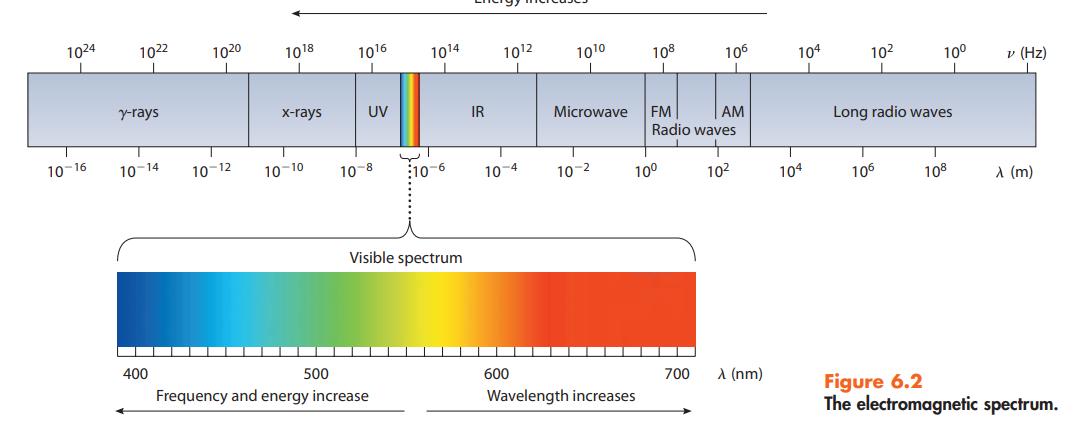
Transcribed Image Text:
1024 10-16 1022 y-rays 10-14 400 1020 10-12 1018 x-rays 10-10 1016 UV 10-8 ان 500 Frequency and energy increase 1014 10-6 Visible spectrum IR 10¹2 10-4 ▬ 600 10¹⁰ Microwave 10-2 108 Wavelength increases 10⁰ FM AM Radio waves 106 700 10² λ (nm) 104 I 104 10² 10⁰ Long radio waves 106 108 v (Hz) A (m) Figure 6.2 The electromagnetic spectrum.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
a In the electromagnetic spectrum as shown in Figure 62 xrays are located to the left of m...View the full answer

Answered By

Akshay Singla
as a qualified engineering expert i am able to offer you my extensive knowledge with real solutions in regards to planning and practices in this field. i am able to assist you from the beginning of your projects, quizzes, exams, reports, etc. i provide detailed and accurate solutions.
i have solved many difficult problems and their results are extremely good and satisfactory.
i am an expert who can provide assistance in task of all topics from basic level to advance research level. i am working as a part time lecturer at university level in renowned institute. i usually design the coursework in my specified topics. i have an experience of more than 5 years in research.
i have been awarded with the state awards in doing research in the fields of science and technology.
recently i have built the prototype of a plane which is carefully made after analyzing all the laws and principles involved in flying and its function.
1. bachelor of technology in mechanical engineering from indian institute of technology (iit)
2. award of excellence in completing course in autocad, engineering drawing, report writing, etc
4.70+
48+ Reviews
56+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Answer the following questions based on the information presented for Cloud 9 in Appendix B of this book and in the current and earlier chapters. You should also consider your answers to the case...
-
Answer the following questions based on two assumptions: (1) Inflation increases the prices of all goods by 20%. (2) Ina's income increases from $50,000 to $55,000. a. Has Ina's budget line become...
-
Answer the following questions based on the information presented for Cloud 9 in Appendix B and in the current and earlier chapters. You should also consider your answers to the case study questions...
-
Fill the blanks of the following table (last column) and find out the value of SDR in terms of U.S. dollars ($/SDR) and the value of U.S. dollars in terms of SDR (SDR/$) The exchange rate is...
-
Solve the following LP problem first graphically and then by the simplex algorithm: Maximize profit = 4X1 + 5X2 Subject to X1 + 2X2 80 3X1 + X2 75 X1, X2 0 What are the values of the basic...
-
On December 31, 2019, Hurly Co. performed environmental consulting services for Cascade Co. Cascade was short of cash, and Hurly Co. agreed to accept a $300,000 zero-interest-bearing note due...
-
Some believe that the top three ratios that every technology company needs to understand and track are the quick ratio, debt to equity ratio, and gross profit margin. Describe each of these ratios...
-
Isolation Company has a debt equity ratio of .80. Return on assets is 7.9 percent, and total equity is $480,000. What is the equity multiplier? Return on equity? Net income? Just Dew It Corporation...
-
These are the pieces you must choose from to summarize and comment on in your essay: "EEOC v. Madison Community Unit School District (No. 12 818 F.2d 577 (7th Cir. 1987))" "Sandor v. Safe Horizon,...
-
Sublimation of 1.0 g of dry ice, CO 2 (s), forms 0.36 L of CO 2 (g) (at 78C and 1.01 10 5 Pa). The expanding gas can do work on the surroundings. Calculate the amount of work done on the...
-
In the lab, you plan to carry out a calorimetry experiment to determine r H for the exothermic reaction of Ca(OH) 2 (s) and HCl(aq). Predict how each of the following will affect the calculated...
-
The paper The Pedaling Technique of Elite Endurance Cyclists (International Journal of Sport Bio mechanics [1991]: 29 53) reported the following data on single-leg power at a high workload: a....
-
1. A nurse manager is discussing the RNs scope of practice with a new hire. Which statement, made by the new hire, is not true of the RNs scope of practice? a.A nurse may be disciplined by the Board...
-
1. A nurse manager is teaching a class about the different role elements of RNs. If she has an adequate understanding, she can state that a care provider is a.a nurse who medically manages patients....
-
Fortune Incorporated is preparing its master budget for the first quarter. The company sells a single product at a price of $25 per unit. Sales (in units) are budgeted at 150,000 for the first...
-
Question: Part One: Memo Write an analysis of the impact of tax laws and incentives as demonstrated in this current years tax return. Specifically, you must address the following: Explain the...
-
An investment advisor currently has two types of investments available for clients: a conservative investment A that pays 6% per year and investment B of higher risk that pays 13%. Clients may divide...
-
1. What are the ways that Folgers is likely to have economies of scale? What are possible sources of diseconomies of scale? 2. Using the product/process matrix, which processes are likely to be used...
-
What are the four types of poultry production systems? Explain each type.
-
Lactones can be prepared from diethyl malonate and epoxides. Diethyl malonate is treated with a base, followed by an epoxide, followed by heating in aqueous acid: Using this process, identify what...
-
Predict the major product of the following transformation. CO2ET C10H100 Heat
-
Consider the structures of the constitutional isomers, Compound A and Compound B (below). When treated with aqueous acid, Compound A undergoes isomerization to give a cis stereoisomer. In contrast,...
-
Assignment 7 The Information Component: Service Desk Performance Measures Part 2 1. What are the four common ways teams measuring their team performance? 2. Suppose you are management person of your...
-
Two drivers (driver A and driver B) are traveling in opposite directions on a long, straight road at different constant velocities. Their motions are represented by the position-time graphs of each,...
-
1. A ball is thrown vertically upward from a window that is 3.6 m above the ground. Its initial speed is 2.8 m/s. With what speed does the ball hit the ground? [4 marks] 2. A 23-kg block is pushed...

Study smarter with the SolutionInn App


