Balance the following equations, and then classify each as a precipitation, acidbase, or gas-forming reaction. (a) KCO3(aq)
Question:
Balance the following equations, and then classify each as a precipitation, acid–base, or gas-forming reaction.
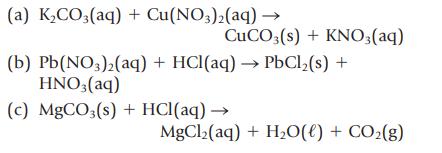
Transcribed Image Text:
(a) K₂CO3(aq) + Cu(NO3)2(aq) → CuCO3(s) + KNO3(aq) (b) Pb(NO3)2(aq) + HCl(aq) → PbCl₂(s) + HNO3(aq) (c) MgCO3(s) + HCl(aq) → MgCl2(aq) + H2O(l) + COz(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
balance and classify each of the given chemical equations a KCOaq CuNOaq CuCOs ...View the full answer

Answered By

Kalyan M. Ranwa
I have more than seven years of teaching experience in physics and mechanical engineering.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Balance the following equations, and then classify each as a precipitation, acidbase, or gas-forming reaction. (a) Ba(OH)(aq) + HCl(aq) BaCl,(aq) + H,O(l) (b) HNO3(aq) + COCO3(s) Co(NO3)2(aq) +...
-
Balance equations for these reactions that occur in aqueous solution, and then classify each as a precipitation, acidbase, or gas-forming reaction. Show states for the products (s, , g, aq), give...
-
Balance the following equations and indicate whether they are combination, decomposition, or combustion reactions: (a) C3H6(g) + O2 (g) CO2 (g) + H2O(g) (b) NH4NO3(s) N2O(g) + H2O(g) (c) C5H6O(I) +...
-
Gems Co. uses the indirect method to prepare its statement of cash flows. The following comparative statement of financial position for 2021 and 2022 are presented: At December 31 2022 2021 Property,...
-
How would you expect a fall in a countrys population to alter its aggregate money demand function? Would it matter if the fall in population were due to a fall in the number of households or to a...
-
In 2012, 15.9% of all U.S. families had incomes below the poverty level, as reported by the U.S. Census Bureau in American Community Survey. During that same year, of 400 randomly selected Wyoming...
-
Explain the following terms related to I.C. engines : (a) Piston speed (b) Compression ratio (c) Brake power (d) Brake thermal efficiency
-
The Speedo LZR Racer Suit is a high-end, body-length swimsuit that was launched on February 13, 2008. When 17 world records fell at the December 2008 European Short Course Championships in Croatia,...
-
You are provided with the following information for Carla Vista Co., effective as of its April 30, 2025, year-end. Accounts payable $844 Accounts receivable 840 Accumulated depreciation-equipment 640...
-
Which two of the following reactions are oxidation reduction reactions? Explain your answer briefly. Classify the remaining reaction. (a) CdCl(aq) + NaS(aq) CdS(s) + 2 NaCl(aq) (b) 2 Ca(s) + O(g) 2...
-
In the following reactions, decide which reactant is oxidized and which is reduced. Designate the oxidizing agent and the reducing agent. 2+ (a) CrO (aq) + 3 Sn+ (aq) + 14 H3O+ (aq) 2 Cr+ (aq) + 3...
-
Is production volume significant in process selection? Explain your answer.
-
What strategies can be employed to enhance visibility and traceability across global supply chains, ensuring compliance with stringent regulatory frameworks and mitigating risks associated with...
-
The pandemic has created significant shifts in consumer demand for certain products. Identify one consumer item that has seen a significant increase. Explain why you believe consumer demand has...
-
Direct marketing is not just plain old direct mail (though it can be). Share an example of when you made a purchase as the result of direct marketing. Be sure to identify the channels that were used...
-
Should cigarette marketers continue to use marketing tactics restricted in one country in other countries where they are not restricted?
-
Superior Company provided the following data for the year ended December 31 (all raw materials are used in production as direct materials): Selling expenses Purchases of raw materials Direct labor...
-
Sogern is an 80%-owned subsidiary of Partplus Company. The two affiliates had the following separate income statements for 2011 and 2012 shown below. Sogern sells at the same gross profit percentage...
-
1. What are some current issues facing Saudi Arabia? What is the climate for doing business in Saudi Arabia today? 2. Is it legal for Auger's firm to make a payment of $100,000 to help ensure this...
-
Assign an IUPAC name for each of the following compounds. a. b. c. d. e. f. g. SH
-
Predict the products that are expected when each of the following compounds is heated with concentrated HBr. a. b. c. d.
-
Draw all constitutionally isomeric ethers with molecular formula C 4 H 10 O. Provide a common name and a systematic name for each isomer.
-
Let's talk about this. Why would the attorney not be subject to discipline for violating the ethical duty of confidentiality if she provides the testimony without objection? Hi there could you help...
-
explain the significance of partition coefficients in determining the distribution of solutes between immiscible phases, and how does this concept guide the choice of solvents in extraction processes...
-
Give your opinion/response to the below- Lukis Anderson arrested December 2012, for murder of Raveesh Kumra but actually it was more of framed by his own DNA. His DNA was found under the finger nail...

Study smarter with the SolutionInn App


