Balance the following equations, and then write the net ionic equation: (a) Zn(s) + HCl(aq) H(g)
Question:
Balance the following equations, and then write the net ionic equation: 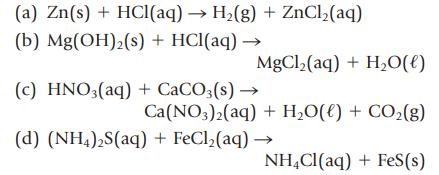
Transcribed Image Text:
(a) Zn(s) + HCl(aq) → H₂(g) + ZnCl₂(aq) (b) Mg(OH)2(s) + HCl(aq) → MgCl₂(aq) + H₂O(l) (c) HNO3(aq) + CaCO3(s) → Ca(NO3)2(aq) + H₂O(l) + CO₂(g) (d) (NH4)2S(aq) + FeCl,(aq) → NH4Cl(aq) + Fes(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a Zns 2HClaq H2g ZnCl2aq Balanced equation Zns 2HClaq H2g ZnCl2aq Net ionic equa...View the full answer

Answered By

Gilbert Chesire
I am a diligent writer who understands the writing conventions used in the industry and with the expertise to produce high quality papers at all times. I love to write plagiarism free work with which the grammar flows perfectly. I write both academics and articles with a lot of enthusiasm. I am always determined to put the interests of my customers before mine so as to build a cohesive environment where we can benefit from each other. I value all my clients and I pay them back by delivering the quality of work they yearn to get.
4.80+
14+ Reviews
49+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) Ba(OH)-(aq) + HPO 4 (aq )- HCIO4 (aq) + Mg(OH )2 (s)
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) CH3COOH (aq) + KOH(aq)- .co.(aq) + NaO H (aq) - HNO3(aq ) + Ba(OH)2(aq )-
-
1) Write balanced net ionic equation for the neutralization ofequal molar amounts of HNO2 and KOH 2) Write balanced net ionic equation for the neutralization ofequal molar amounts of HBr and NH3 3)...
-
Your independent film production company owns the rights to a script for a rom-com movie. They estimate that the script would cost $30 million to make into a movie, which would have a 35% chance of...
-
Using the data in the Memoranda to Table calculate the U.S. 2009 net international investment position with direct investments valued at marketprices. International Investment Position of the United...
-
A computer repair shop has two work centers. The first center examines the computer to see what is wrong, and the second center repairs the computer. Let x 1 and x 2 be random variables representing...
-
Air-fuel ratio in petrol engine is controlled by (a) fuel pump (b) injector (c) controller (d) carburettor
-
Selected cost data for Antique Poster Co. are as follows: Estimated manufacturing overhead cost for the year....$ 120,000 Estimated direct labor cost for the year......... 100,000 Actual...
-
Would you be able to recreate the planning and strategic management process that Mr. Nadella used? What would Microsoft's planning flowchart look like based on the goal he proposed? Did Mr. Nadella...
-
Write two chemical equations, one that shows H 2 O reacting (with HBr) as a Brnsted base and a second that shows H 2 O reacting (with NH 3 ) as a Brnsted acid.
-
Write an equation that describes the equilibrium that exists when the weak acid benzoic acid (C 6 H 5 CO 2 H) dissolves in water. Identify each of the four species in solution as either Brnsted acids...
-
The article "Analysis of Unwanted Fire Alarm: Case Study" (W. Chow, N. Fong, and C. Ho, Journal of Architectural Engineering, 1999:62-65) presents a count of the number of false alarms at several...
-
How to protect your ownership of the company you founded? Given the case of purchasing 9.2% stake of TWTR?
-
Assume you are preparing for a job interview as a social service worker. And you were asked these questions during the job interview. Answer the following questions to the best of your understanding....
-
Explain the phenomena of 'overinvestment' and 'underinvestment' in relation to a firm's investment strategy and its relationship to NPV. In each case give a reason as why a firm or its management...
-
(b) What risk factors might pertain to a private equity investment in the pandemic? (9 marks) (c) Explain how a MBO deal is usually structured both in terms of its corporate/legal structure and its...
-
(a) Explain the difference between a seed investment and a start-up investment and how they compare on risk considerations. (6 marks) (b) Both of these stages of investment are often financed...
-
Refer to the preceding information for Fast Cools acquisition of Fast Airs common stock. Assume Fast Cool issues 40,000 shares of its $20 fair value common stock for 100% of Fast Airs common stock....
-
Government is advised to tax goods whose demand curves are inelastic if the goal is to raise tax revenues. If the goal is to discourage consumption, then it ought to tax goods whose demand curves are...
-
Identify the reactants that you would use to make each of the following imines: (a) (b) (c) N.
-
Predict the product of each of the following reactions: (a) (b) [1] -NH2 -H20 [H*] H2N-NH2 -H20
-
Identify the reactants that you would use to make each of the following compounds: (a) (b) NH2
-
An office building has three floors of rentable space with a single tenant on each floor. The first floor has 20,000 square feet of rentable space and is currently renting for $15 per square foot....
-
Explain what is an organizational code of ethics (Ch 3 topic)? Why should organizations have a code of ethics? Explain the "three nudge systems" discussed in the media clip. Explain why or why not,...
-
How would you describe a highly effective leader in data science environment? Why?

Study smarter with the SolutionInn App


