Calculate equilibrium constants for the following reactions at 298 K. Indicate whether the equilibrium as written is
Question:
Calculate equilibrium constants for the following reactions at 298 K. Indicate whether the equilibrium as written is reactant- or product-favored at equilibrium. 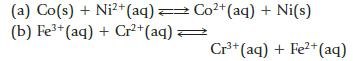
Transcribed Image Text:
Co²+ (aq) + Ni(s) (a) Co(s) + Ni²+ (aq) = (b) Fe³+ (aq) + Cr²+ (aq) → Cr³+ (aq) + Fe2+ (aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a Cos Niaq Coaq Nis The standard reduction potential for Coaq to Cos is 028 V and the standard reduc...View the full answer

Answered By

Madhur Jain
I have 6 years of rich teaching experience in subjects like Mathematics, Accounting, and Entrance Exams preparation. With my experience, I am able to quickly adapt to the student's level of understanding and make the best use of his time.
I focus on teaching concepts along with the applications and what separates me is the connection I create with my students. I am well qualified for working on complex problems and reaching out to the solutions in minimal time. I was also awarded 'The Best Tutor Award' for 2 consecutive years in my previous job.
Hoping to get to work on some really interesting problems here.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Calculate equilibrium constants for the following reactions at 298 K. Indicate whether the equilibrium as written is reactant- or product-favored at equilibrium. (a) 2 Cl(aq) + Br(e) (b) Fe+ (aq) +...
-
Calculate the equilibrium constants of the following reactions at 25C from standard potential data: (a) Sn(s) + CuS04 (aq) ~ Cu(s) + SnS04 (aq) (b) Cu2+(aq) + Cu(s) ~ 2 Cu+{aq)
-
Predict whether the equilibrium constants for the following reactions should be greater than 1 or less than 1: (a) CdI(s) + CaF(s) CdF(s) + Cal,(s) (b) [Cul 1-(aq) + [CuCl](aq) (c) NH;(aq)...
-
You accepted a new job with starting salary of $52,000 per year. The salary is expected to increase 4% each year. Now it is time to make a retirement plan for the next 39 years you expect to work....
-
Carnival Corporation has recently placed into service some of the largest cruise ships in the world. One of these ships, the Carnival Glory, can hold up to 3,000 passengers and cost $530 million to...
-
Thunder Attractions, an amusement park, is considering a capital investment in a new exhibit. The exhibit would cost 136,000 and have an estimated useful life of 5 years. It will be sold for 60,000...
-
The probability of randomly choosing a cricket player who also played for his school team. (Assume that you are choosing from the population of all cricket players.) Write a statement that represents...
-
Drive and Fly, near an airport, incurred the following costs to acquire land, make land improvements, and construct and furnish a small building: a. Purchase price of three acres of land $ 80,000 b....
-
Lets say a portion of money that you add, usually, in a package at Quellenhof to cover the fee. Example: a hotel room costs 200 and the flight 100. Total 300. However, the guest did not book it...
-
Two Ag + (aq) | Ag(s) half-cells are constructed. The first has [Ag + ] = 1.0 M, the second has [Ag + ] = 1.0 10 5 M. When linked together with a salt bridge and external circuit, a cell potential...
-
A voltaic cell set up utilizing the reaction has a cell potential of 0.45 V at 298 K. Describe how the potential of this cell will change as the cell is discharged. At what point does the cell...
-
Marthas RRSP is currently worth $97,000. She plans to contribute $5000 at the beginning of every six months until she reaches age 58, 12 years from now. Then she intends to use half of the funds in...
-
Writing is one of the essential skills at school. When learners struggle to write, teachers get frustrated. Describe how you can identify learners who experience barriers to writing. Specific...
-
what ways does the novelist orchestrate a multifaceted conflict landscape, interweaving interpersonal, intrapersonal, and societal conflicts to create a narrative tapestry that reflects the...
-
How does accounting differ from budgeting? 2. Why are principles of accounting so important? 3. Who uses nonprofits' financial information and why? 4. Give an example of cash accounting, accrual...
-
.How does the playwright employ the dialectical tension between tradition and modernity to evoke a sense of existential conflict among the characters, ultimately interrogating the socio-cultural...
-
What is constitutional law in the U.S? Where did it originate? By what right are laws made? Who interprets laws? How has seminal constitutional and contractual case law and the influence of these on...
-
A researcher convinced a restaurant to experiment with two different kinds of music. One was faster upbeat music and the second was soft relaxing music. A random sample of diners was drawn and the...
-
Don Griffin worked as an accountant at a local accounting firm for five years after graduating from university. Recently, he opened his own accounting practice, which he operates as a corporation....
-
In the following Verilog process A, B, C, and D are all registers that have a value of 0 at time = 10ns. If E changes from 0 to 1 at time = 20ns, specify the time(s) at which each signal will change...
-
In the following Verilog process A, B, C, and D are all registers that have a value of 0 at time = 10 ns. If E changes from 0 to 1 at time = 20 ns, specify the time(s) at which each signal will...
-
Draw the hardware obtained if the following code is synthesized: module reg3 (Q1,Q2,Q3,Q4, A,CLK); input A; input CLK; output Q1,Q2,Q3,Q4; reg Q1,Q2,Q3,Q4; always @(posedge CLK) begin Q4 = Q3; Q3 =...
-
1) Entity J had an accounts receivable turnover ratio of 8 times in 2024 and 6 times in 2025. From 2024 to 2025, the accounts receivable turnover ratio: Entity J had an accounts receivable turnover...
-
Imagine you've just stepped into the shoes of a top financial detective! Your latest case? Unraveling the enigmatic story behind the numbers of Slater Inc., a trendy retail powerhouse making waves in...
-
Grady Inc. owns 100% of the shares of Plaza Ltd. Grady has decided that it does not want Plaza to continue as a separate legal entity. It has asked your assistance on whether to absorb Plaza into its...

Study smarter with the SolutionInn App


