The average energy output of a good grade of coal is 2.6 10 7 kJ/ton. Fission
Question:
The average energy output of a good grade of coal is 2.6 × 107 kJ/ton. Fission of 1 mol of 235U releases 2.1 × 1010 kJ. Find the number of tons of coal needed to produce the same energy as 1 lb of 235U. (See Appendix C for conversion factors.)
Data given in Appendix C
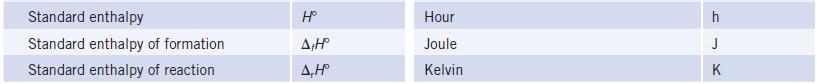
Transcribed Image Text:
TABLE 3 Some Common Abbreviations and Standard Symbols Abbreviation Term Activation energy Ampere Aqueous solution Atmosphere, unit of pressure Atomic mass unit Avogadro's constant Bar, unit of pressure Body-centered cubic Bohr radius Boiling point Celsius temperature Charge number of an ion Coulomb, electric charge Curie, radioactivity Cycles per second, hertz Debye, unit of electric dipole Electron Electron volt Electronegativity Energy Enthalpy E₂ A aq atm u N bar bcc a bp °C Z C Ci Hz D e eV XEH Term Entropy Standard entropy Entropy change for reaction Equilibrium constant Concentration basis Pressure basis lonization weak acid lonization weak base Solubility product Formation constant Ethylenediamine Face-centered cubic Faraday constant Gas constant Gibbs free energy Standard free energy Standard free energy of formation Standard free energy change for reaction Half-life Heat Hertz Abbreviation S Sº 4,Sº K K Kp K₂ Kb Ksp K₁ en fcc F R G Gº A,Gº A,Gº tuz 9 Hz
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
Convert the energy output of 1 lb of 235U to kJ 1 lb 453592 g E 21 1010 kJmol 453592 glb 1 mol...View the full answer

Answered By

Ritvik Mahajan
I scored 98,98,97 in my Class 12 final Exams in Physics , Chemistry and Mathematics. I have an aggregate of 9.13 in the five semesters of my Bachelor of Technology(CS) Degree. No teaching experience as such but i help students in my neighborhood solve problems. I love problem solving and it would be great to help you guys out
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
The thermite reaction is a very exothermic reaction; it has been used to produce liquid iron for welding. A mixture of 2 mol of powdered aluminum metal and 1 mol of iron(III) oxide yields liquid iron...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
A small atomic bomb releases energy equivalent to the detonation of 20,000 tons of TNT; a ton of TNT releases 4 X 109 J of energy when exploded. Using 2 X 1013 J/mol as the energy released by fission...
-
Suppose that there are two countries, A and B, and two goods, X and Y. Country A has a production possibility frontier (PPF) that is given by the equation X + Y = 100, while country B has a PPF that...
-
Juras Inc. and Hinson Inc. have the following operating data: a. Compute the operating leverage for Juras Inc. and Hinson Inc. b. How much would income from operations increase for each company if...
-
A pinhole of diameter \(0.20 \mathrm{~mm}\) is illuminated with \(550 \mathrm{~nm}\) light. What is the width of the Airy disk on a screen \(1.5 \mathrm{~m}\) away?
-
What is Artificial Life? How does it relate to Artificial Intelligence? Is one an alternative to the other, or are they complementary?
-
Camus Blalack, process engineer, knows that the acceptance of a new process design will depend on its economic feasibility. The new process is designed to improve environmental performance. On the...
-
6. A thin rod with length L is placed along the y-axis as shown in the figure. The midpoint of the rod is at the origin. The rod has been given a non-uniform charge density given by = 4(y+d) y dq P...
-
The age of minerals can sometimes be determined by measuring the amounts of 206 Pb and 238 U in a sample. This determination assumes that all of the 206 Pb in the sample comes from the decay of 238...
-
Sodium-23 (in a sample of NaCl) is subjected to neutron bombardment in a nuclear reactor to produce 24 Na. When removed from the reactor, the sample is radioactive, with activity of 2.54 10 4 dpm....
-
The Metro Packaging Company in Edmonton produces clear plastic bottles for the Kooler Cola Company, a soft-drink manufacturer. Metro inspects each lot of 5000 bottles before they are shipped to the...
-
What advice would you give someone who is actively seeking work in relation to their activities on social media? What should they avoid saying or doing? Should they consider tidying up their...
-
ChoicePoint is the largest company in the $4 billion background-checking industry. Is there anything ChoicePoint can do, as the industry leader, to be more sensitive and respectful to the privacy...
-
Form a small group with several class members and discuss the following scenario: The owner-manager of a small, four-person consulting firm works long hours on multiple projects and expects his three...
-
As described in the case on New Belgium Brewing Company as well as in this chapters opening vignette, companies are coming up with creative ways to introduce fun into the workplace in order to...
-
A British Workplace Behavior Survey used a sample of 4,000 employees from four large organizations, and discovered three broad categories of ill-treatment at work. These included violence and injury...
-
Miller Minerals Co. manufactures a product that requires the use of a considerable amount of natural gas to heat it to a desired temperature. The process requires a constant level of heat, so the...
-
A simple random sample of 220 university students were asked what pasta they usually order and with which sauce. The preferences of these respondents are summarised below: Sauce Bolognese Pasta...
-
Repeat Problem 6.97 if the tank is sealed and a pressure of 5.0 psig is above the water in the tank. Repeat Problem Compute the time required to empty the tank shown in Fig. 6.14 if the original...
-
Compute the time required to reduce the depth in the tank shown in Fig. 6.14 by 21.0 ft if the original depth is 23.0 ft. The tank diameter is 46.5 ft and the orifice diameter is 8.75 in. dh
-
Compute the time required to reduce the depth in the tank shown in Fig. 6.14 by 12.5 in if the original depth is 38 in. The tank diameter is 6.25 ft and the orifice diameter is 0.625 in. dh
-
Using the following information, calculate total assets assuming all accounts have normal balances. Accounts payables..........$1500 Fees earned.. .$3600 Accounts receivables......1800 Utility...
-
4. What measure of location provides a measure of central location computed by summing the data and dividing by the number of observations?
-
In the case of the stock price went up to 65 to 40, your current margin increased to 81.5%, what is the amount you can buy without additional equity

Study smarter with the SolutionInn App


