The equation for the reaction of phosphorus and chlorine is If you use 8000 molecules of P
Question:
The equation for the reaction of phosphorus and chlorine is ![]() If you use 8000 molecules of P4 in this reaction how many molecules of Cl2 are required to consume the P4 completely?
If you use 8000 molecules of P4 in this reaction how many molecules of Cl2 are required to consume the P4 completely?
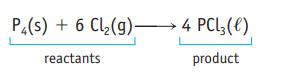
Transcribed Image Text:
P4(s) + 6 Cl₂(g) → 4 PCl3(l)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (5 reviews)
The balanced chemical equation provided is P4s 6 Cl2g 4 PCl3l In this equation e...View the full answer

Answered By

Raunak Agarwal
Teaching is my hobby and now my profession. I teach students of CA and CFA(USA) in batches of 100 students and have a 5 year experience.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Use the subset of 401KSUBS.RAW with fsize = 1; this restricts the analysis to single person households; see also Computer Exercise C4.8. (i) What is the youngest age of people in this sample? How...
-
Use the estimator in the myfico website to estimate the FICO credit score for the following person: -Never had a budget. Not sure where his money is going. Regularly uses credit cards to pay for...
-
The problem requires you to use File C03 on the computer problem spreadsheet. Diction Publishing estimates that it needs $500,000 to support its expected growth. The underwriting fees charged by the...
-
Two slot machines offer to double your money 3 times out of 5. Machine A takes $10 bets and Machine B takes $100 bets on each occasion. A risk-averse investor prefers to bet on A) Machine A B)...
-
For a small country like the Philippines, a move to free trade would have huge advantages. It would let consumers and producers make their choices based on the real costs of goods, not artificial...
-
Refer to all of the facts in Problem 11-1A. Problem 11-1A. Kebler Company was incorporated as a new business on January 1, 2017. The corporate charter approved on that date authorized the issuance of...
-
Water is raised to a height of 40 m by means of a centrifugal pump. If the quantity of water raised is 350 m3/h, determine the power of electric motor required to drive the pump. Take efficiency of...
-
Larned Corporation recorded the following transactions for the just completed month. a. $ 80,000 in raw materials were purchased on account. b. $ 71,000 in raw materials were requisitioned for use in...
-
Which do you think are more important to emphasize in a literature review, the opinions of experts in the field or related studies and why? Describe the kind of behaviours that individual team...
-
Which of the following compounds has the highest mass percent of chlorine? (a) BCl 3 (b) AsCl 3 (c) GaCl 3 (d) AlCl 3 (e) PCl 3
-
Write an equation from the following description: reactants are gaseous NH 3 and O 2 , products are gaseous NO 2 and liquid H 2 O, and the stoichiometric coefficients are 4, 7, 4, and 6,...
-
The Malon Appliance Co. provides a 120-day parts-and-labor warranty on all merchandise it sells. Malon estimates the warranty expense for the current period to be $2,450. During this period, a...
-
In the air duct system shown in the figure, air enters the main duct at A. The recommended Fan velocities in the main duct sections and branches are 10 m/s and 7 m/s, respectively. Using the velocity...
-
The management of Elite Limited is struggling to understand changes in their financial performance and the trends emerging within their accounting data. Hence, the company would like to know about...
-
On January 1 of Year 1, Innovative Lab issued a 4-year $50,000 note to a local bank with fixed interest payments based on 6%, payable annually on December 31. To hedge the risk of a fixed interest...
-
Describe the operating activities of each company noting similarities and difference between COCA COLA & PEPSICO. Identify two economy wide factors and industry wide factors that could impact on the...
-
Macy Pharmacy has a project which has the following cash flows. Year 0 -$200,000 Year 1 = $50,000 Year 2 $100,000 Year 3 $150,000 Year 4 $40,000 = Year 5 $25,000 = The cost of capital is 10%. What is...
-
What is meant by date alignment? Does it exist on the consolidated worksheet under the following methods, and if not, how is it created prior to elimination of the investment account under each of...
-
Suppose the S&P 500 futures price is 1000, = 30%, r = 5%, = 5%, T = 1, and n = 3. a. What are the prices of European calls and puts for K = $1000? Why do you find the prices to be equal? b. What...
-
For each of the compounds below, locate the lone pair adjacent to a positive charge and draw the resonance structure: a. b. c. N.
-
Assume the following simplified dependence of the pressure in a ventricle of the human heart as a function of the volume of blood pumped. P s , the systolic pressure, is 120. mm Hg, corresponding to...
-
Electrical current is passed through a resistor immersed in a liquid in an adiabatic container. The temperature of the liquid is varied by 1C. The system consists solely of the liquid. Does heat or...
-
Category Land Land improvements Buildings Plant Asset $ 165,000 and Amortization $ 1,000,000 219,251 Equipment 625,000 307,500 Automobiles and trucks 162,000 90,325 Leasehold improvements 196,000...
-
Given that 57.7 fex is equal to 1 gal and that 14.3 glo is equal to 1 day, how many cL/hr are there in 0.568 fex/glo? ( NOTE: A fex and glo are are both fictitious units of volume and time,...
-
For shooting practice a person uses a pellet gun and an empty pop can. The pop can rests on a flat surface that has a coefficient of kinetic friction 0.500. The pellet has a mass of 0.120 kg and the...

Study smarter with the SolutionInn App


