Write an equation from the following description: reactants are gaseous NH 3 and O 2 , products
Question:
Write an equation from the following description: reactants are gaseous NH3 and O2, products are gaseous NO2 and liquid H2O, and the stoichiometric coefficients are 4, 7, 4, and 6, respectively.
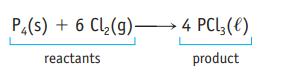
Transcribed Image Text:
P4(s) + 6 Cl₂(g) 4 PCL3 (1) reactants product
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
The balanced chemic...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
The idea is to get an equation from the vanishing of the determinant of a homogeneous linear system as the condition for a nontrivial solution in Cramers theorem. We explain the trick for obtaining...
-
Jerry Clark is the sole proprietor of Affordable Digital Hearing, which does business by repairing hearing aids. In June 2006, Clark received a call from Kevin Wilson, a representative of...
-
The Chief Financial Officer at Ford Motor Company is said to usea hybrid-costing system. Define the hybrid-costing system. Explainthe advantages to this company to use this system. I want a 10 page 2...
-
Suppose that workers involved in manufacturing are paid less than all other workers in the economy. What would be the effect on the real income distribution within the economy if there were a...
-
COMMON CORE SMP MATERIALS GROUPING GETTING STARTED Example: 5)167 Solution: H. Develop the division algorithm and reinforce the partitioning model and the place-value concepts associated with...
-
A single acting reciprocating pump has to deliver water against a total head of 50 m. The plunger diameter is 120 mm. What is the force exerted by the plunger during delivery stroke? Assume pump...
-
Forham Inc. manufactures stainless steel knives. Following are factory costs incurred during 2010: Material costs Stainless steel ............... $800,000 Equipment oil and grease .......... 6,000...
-
Explain the role of policy in addressing the injustice in community health facilities.?
-
The equation for the reaction of phosphorus and chlorine is If you use 8000 molecules of P 4 in this reaction how many molecules of Cl 2 are required to consume the P 4 completely? P4(s) + 6 Cl(g) 4...
-
Strontium has four stable isotopes. Strontium-84 has a very low natural abundance, but 86 Sr, 87 Sr, and 88 Sr are all reasonably abundant. Knowing that the atomic weight of strontium is 87.62, which...
-
Refer to E16.19. If Worth Company produced 100,000 barrels of oil in its first year, what would be the accumulated depletion balance? Data from E16.19 Worth Company recently purchased an oil well for...
-
43. A simple pendulum has a length of 1.20 m and is pulled from equilibrium by 4.00 to the right and then released. The clock starts (t = 0) when the mass on the simple pendulum is at a position of...
-
Calculating Present Values Imprudential, Inc., has an unfunded pension liability of $645 million that must be paid in 25 years. To assess the value of the firm's stock, financial analysts want to...
-
Addressing the following questions is part of HR strategy Except. a . Does the HR department offer their employees competencies that are required to meet their goals? b . What are the steps that a...
-
A student is running at her top speed of 5.5 m/s to catch a bus, which is stopped at the bus stop. When the student is still a distance 38.5 m from the bus, it starts to pull away, moving with a...
-
If you were asked whether a large university such as Tennessee or Michigan with a large seating capacity for their football stadiums should build a new football stadium, how would you respond and...
-
A parent company paid $500,000 for a 100% interest in a subsidiary. At the end of the first year, the subsidiary reported net income of $40,000 and paid $5,000 in dividends. The price paid reflected...
-
The time to assemble the first unit on a production line is 10 hours. The learning rate is 0.94. Approximately how long will it take for the seventh unit to be assembled? The number of hours needed...
-
Two ideal gas systems undergo reversible expansion under different conditions starting from the same P and V. At the end of the expansion, the two systems have the same volume. The pressure in the...
-
You have a liquid and its gaseous form in equilibrium in a piston and cylinder assembly in a constant temperature bath. Give an example of a reversible process.
-
Describe how reversible and irreversible expansions differ by discussing the degree to which equilibrium is maintained between the system and the surroundings.
-
Berset Inc. (BI) has just issued semi-annual coupon bonds with 12 years to maturity and a face value of $1,000 per bond. The bonds make semi-annual coupon payments of $60. What is the price of one...
-
You plan to invest $10,000 into your new savings account the end of the year for the next 10 years. The interest earned in the account is 8% per year. How much do you expect to have in the account at...
-
Duggins Veterinary Supplies can issue perpetual preferred stock at a price of $50 a share with an annual dividend of $4.50 per share. Duggins must pay flotation costs of 5% of the issue price. What...

Study smarter with the SolutionInn App


