The equilibrium constant, K p , for N 2 O 4 (g) 2 NO 2 (g)
Question:
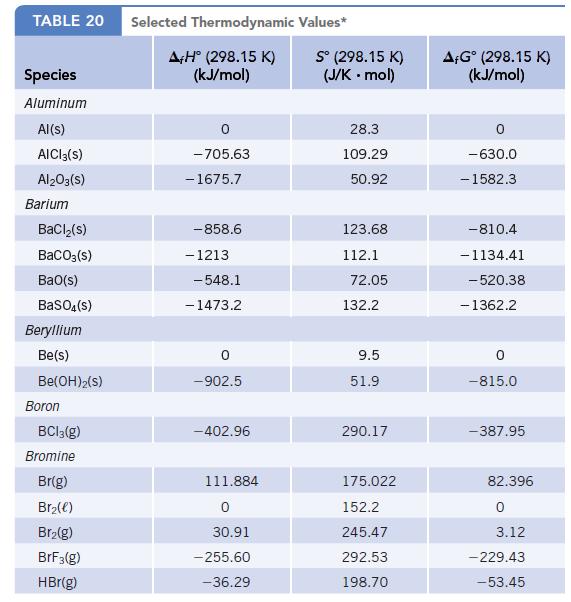
The equilibrium constant, Kp, for N2O4(g) ⇄ 2 NO2(g) is 0.14 at 25°C. Calculate ΔrG° for the conversion of N2O4(g) to NO2(g) from this constant, and compare this value with that determined from the Δf G° values in Appendix L.
Data given in Appendix L
Transcribed Image Text:
TABLE 20 Species Aluminum Al(s) AICI 3(S) Al2O3(S) Barium BaCl₂(s) BaCO3(s) BaO(s) BaSO4(s) Beryllium Be(s) Be(OH)2(S) Boron BC13(g) Bromine Br(g) Br₂(e) Br₂(g) BrF3(g) HBr(g) Selected Thermodynamic A+Hº (298.15 K) (kJ/mol) -705.63 - 1675.7 -858.6 - 1213 -548.1 -1473.2 0 -902.5 -402.96 111.884 0 30.91 -255.60 -36.29 Values* Sº (298.15 K) (J/K.mol) 28.3 109.29 50.92 123.68 112.1 72.05 132.2 9.5 51.9 290.17 175.022 152.2 245.47 292.53 198.70 AFGᵒ (298.15 K) (kJ/mol) -630.0 - 1582.3 -810.4 -1134.41 -520.38 - 1362.2 0 -815.0 -387.95 82.396 0 3.12 -229.43 -53.45
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
To calculate rG the standard Gibbs free energy change for the conversion of N2O4g to 2 NO2g using th...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Calculate the standard free-energy change and the equilibrium constant Kp for the following reaction at 25oC. See Appendix C for data. CO(g) + 2H2(g) =CH,OH(g)
-
The equilibrium constant KP for the reaction is 2 Ã 10-42 at 25°C. (a) What is Kc for the reaction at the same temperature? (b) The very small value of KP (and Kc) indicates that the...
-
Calculate the standard free-energy change and the equilibrium constant Kp for the following reaction at 25 C. See Table 18.2 for data. CO(g) 3H2(g) CH4(g) H20(g)
-
The current quoted price of a 13% coupon bond is $110. It pays coupon semi-annually. The next coupon will be paid in 6-days (total number of days in this semi-annual period is 181) and the futures...
-
In what ways might classification be useful in any field of study? Use international differences in financial reporting as an illustration of your answer.
-
The article ??The Ball-on-Three-Ball Test for Tensile Strength: Refined Methodology and Results for Three Hohokam Ceramic Types?? (M. Beck, American Antiquity, 2002:558??569) discusses the strength...
-
Qin Ltd wants to determine its cost of capital to use in future capital budgeting decisions. The companys capital structure is as follows. Required (a) Calculate the companys cost of capital based on...
-
For each of the following inventory errors occurring in 2013, determine the effect of the error on 2013s cost of goods sold, net income, and retained earnings. Assume that the error is not discovered...
-
Applying analytical synthesis of mechanisms, design a four-bar mechanism to move the object shown in the figure, through the three positions indicated in their numbered order without taking into...
-
Elemental boron, in the form of thin fibers, can be made by reducing a boron halide with H 2 . BCl 3 (g) + 32 H 2 (g) B(s) + 3 HCl(g) Calculate r H, r S, and r G at 25C for this reaction. Is the...
-
Sodium reacts violently with water according to the equation Na(s) + H 2 O() NaOH(aq) + 12 H 2 (g) Without doing calculations, predict the signs of r H and r S for the reaction. Verify your...
-
Your company has been approached to bid on a contract to sell 18,000 voice recognition (VR) computer keyboards per year for four years. Due to technological improvements, beyond that time they will...
-
What is the relationship between the price of a financial asset and the payments investors will receive from owning that asset?
-
Describe briefly the respective uses of demand drafts, sight drafts, and time drafts in commercial practice.
-
Define the term required reserve ratio.
-
What is the greater fool theory?
-
What is the difference in legal effect between a drawer having her cheque certified and a payee later having the cheque certified?
-
Refer to the information for Coves Cakes in E6-3. Price per cake ....... $17.00 Variable cost per cake Ingredients ........ 2.50 Direct labor ....... 1.40 Overhead (box, etc.) ..... 0.20 Fixed cost...
-
Imagine you are the HR manager at a company, and a female employee came to you upset because she felt a male coworker was creating a hostile work environment by repeatedly asking her out on dates...
-
A simulated infrared absorption spectrum of a gas-phase organic compound is shown in the following figure. Use the characteristic group frequencies listed in Section 19.5 to decide whether this...
-
Isotopic substitution is used to identify characteristic groups in an unknown compound using vibrational spectroscopy. Consider the C~C bond in ethane ( 12 C 2 1 H 6 ). By what factor would the...
-
A simulated infrared absorption spectrum of a gas-phase organic compound is shown in the following figure. Use the characteristic group frequencies listed in Section 19.5 to decide whether this...
-
The compound interest formula is below, where R is the future value of the investment, r is the annual interest rate (as a decimal), n is the number of times interest is compounded each year, t is...
-
How does synergistic cooperation manifest in intricate interdisciplinary projects requiring specialized expertise ? Explain
-
Sally has highlighted concerns that Lakeside is susceptible to a risk of sending tow vehicles to jobs that do not exist. This is because the business has yet to develop a reliable way of validating...

Study smarter with the SolutionInn App


