The mass spectrum of nitrogen dioxide is illustrated here. (a) Identify the cations present for each of
Question:
The mass spectrum of nitrogen dioxide is illustrated here.
(a) Identify the cations present for each of the four peaks in the mass spectrum.
(b) Does the mass spectrum provide evidence that the two oxygen atoms are attached to a central nitrogen atom (ONO), or that an oxygen atom is at the center (NOO)? Explain.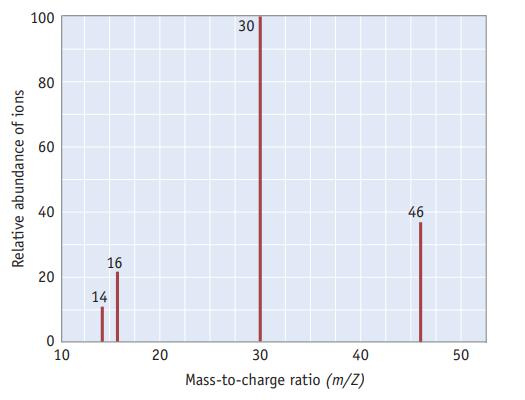
Transcribed Image Text:
Relative abundance of ions 100 80 60 40 20 10 16 14 20 30 30 40 Mass-to-charge ratio (m/Z) 46 50
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a The four peaks in the mass spectrum correspond to the following cations Pe...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
The source of oxygen that drives the internal combustion engine in an automobile is air. Air is a mixture of gases, principally N2 (~79%) and O2 (~20%). In the cylinder of an automobile engine,...
-
The mass spectrum of unknown compound A shows a molecular ion at m/z 116 and prominent peaks at m/z 87 and m/z 101. Its UV spectrum shows no maximum above 200 nm. The IR and NMR spectra of A follow....
-
Mass spectrometry is more often applied to molecules than to atoms. We will see in Chapter 3 that the molecular weight of a molecule is the sum of the atomic weights of the atoms in the molecule. The...
-
solve system of equation: 22-4=4 81+y = -9 @solve the system of equation: x-Y = -5 xy = -3 find larger and smuller value and intersection point. 3) Solve following system of equation 2 x+y=0- 2 x -...
-
What types of documentation does a systems analyst prepare, and what would be included in each type?
-
Based on the chart of betas in Figure 10-16 How would you describe the relative riskiness of investing in Bank of America? Procter & Gamble Co Kraft Foods Inc. McDonald's Corp Merck & Co. Inc....
-
Consider the flowsheet for the manufacture of vinyl chloride in Figure 2.6. (a) If the pyrolysis furnace and distillation towers are operated at low pressure \((1.5 \mathrm{~atm})\), what are the...
-
Platinum Companys inventory of XL125 on January 1 was 8,000 gallons, costing $ 0.45 per gallon (periodic inventory). In addition to this beginning inventory, purchases during the next six months were...
-
As you lead your organization's performance management efforts, your chief human resources officer has real concerns about the buy-in among your staff. She has come to you to share her thoughts. With...
-
The mass spectrum of phosphoryl chloride, POF 3 , is illustrated here. (a). Identify the cation fragment at a m/Z ratio of 85. (b). Identify the cation fragment at a m/Z ratio of 69. (c). Which two...
-
Epsom salt is used in tanning leather and in medicine. It is hydrated magnesium sulfate, MgSO 4 7 H 2 O. The water of hydration is lost on heating, with the number lost depending on the temperature....
-
Helmuth Inc's latest net income was $1,210,000, and it had 225,000 shares outstanding. The company wants to pay out 45% of its income. What dividend per share should it declare? a. $2.49 b. $2.06 c....
-
In 1929, U.S. GDP was, adjusted for inflation, $1,056.6 billion. The following year it was, also adjusted for inflation, $966.7 billion. What was the growth rate of U.S. in 1930? (Do not type the...
-
Table of Interest Rates: Time Period Oyly Oy2y 0v3y lyly 2yly Consider the following bond: Exactly 3 years to maturity 9.7% coupon rate, paid annually $100 par value Bond is callable in exactly 1...
-
An amount of $13,500.00 is deposited into an account today, it is expected to mcrease to a maturity value of $15,349.34 in 6.5 years from now. What is the nominal interest rate compounded quarterly?...
-
Royal Lawncare Company produces and sells two packaged products Weed ban and Green grow. Revenue and cost information relating to the products follow: Product Weed ban Green grow Selling price per...
-
A 50 cm x 30 cm x 20 cm block weighing 150 N should be moved at a constant speed of 0.8 m/s up an oblique plan with a coefficient of friction f= 0.27. a) Determine the force F required in the...
-
XCV, Inc., which manufactures automobile parts for assembly, is considering the costs and the benefits of leverage. The CFO notes that the return on equity of the firm, which is only 12.75% now based...
-
Test whether the 5-year survival rate for breast cancer is significantly different between African American and Caucasian women who are younger than 50 years of age and have localized disease....
-
In each of the following cases draw the structure of an alkyl halide that will undergo an E2 elimination to yield only the indicated alkene. a. b. c. d. ? = E2 E2
-
Consider the following reaction: a. Draw the mechanism of this reaction. b. What is the rate equation of this reaction? c. Draw an energy diagram of the reaction. H2SO, Heat OH
-
Equation (3.38), C P = C V + TV ( 2 /). links C P and C V with and . Use this equation to evaluate C P C V for an ideal gas.
-
11. Let A= -237---8- and = -H] Suppose and v are eigenvectors of A. Use this information to diagonalize A.
-
3. Let (S, o) be a semigroup. If for x, y S, xoy = y = yox, prove that (S,o) is an abelian group.
-
Simplify 516x11 completely. Write answer in radical form. Assume Answer = Entry Tip: To enter an answer like 50x4x2, type 50x^4*root(3) answer before submitting! Question Help: Message instructor...

Study smarter with the SolutionInn App


