The solid-state structure of silicon carbide is shown below. (a) How many atoms of each type are
Question:
The solid-state structure of silicon carbide is shown below.
(a) How many atoms of each type are contained within the unit cell? What is the formulas of silicon carbide?
(b) Knowing that the Si—C bond length is 188.8 pm (and the Si—C—Si bond angle is 109.5°), calculate the density of SiC.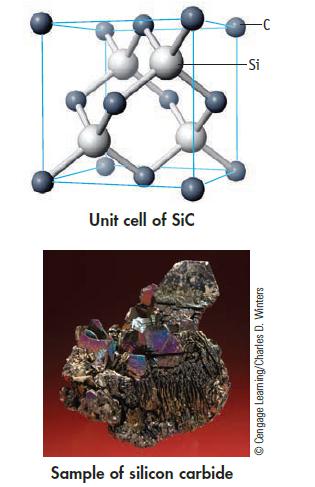
Transcribed Image Text:
Unit cell of Sic Sample of silicon carbide -Si Ⓒ Cengage Learning/Charles D. Winters
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
a In the unit cell of silicon carbide SiC there are four silicon Si atoms and four carbon C atoms Ea...View the full answer

Answered By

Umber Talat
I am providing full time mentoring and tutoring services in Business Finance, Contemporary issue in Global Economy, Quantitative Techniques, Principles of Marketing, strategic marketing, International Marketing, Organizational Behavior (OB), Consumer Behavior, Sales Force Management, Strategic Brand Management, Services Marketing, Integrated Marketing Communication (IMC), Principles of Management, General Management, Strategic Management, Small and Medium Enterprise Management, Innovation Management, Change Management, Knowledge Management, Strategic Planning, Operations Management, Supply Chain Management, Logistics Management, Inventory management, Total Quality Management (TQM), Productions Management, Project Management, Production Planning, Human Resource Management (HRM), Human Resource Development, Strategic HRM, Organizational Planning, Performance and Compensation Management, Recruitment and Selection, Organizational Development, Global Issues in Human Resource Management, Retail Marketing, Entrepreneurship, Entrepreneurial Marketing, International Business, Research Methods in Business, Business Communication, Business Ethics.
4.70+
158+ Reviews
236+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
(a) Figure 22.37 shows eight corner-sharing ReO 6 octahedra in the solid state structure of ReO 3 . From this, derive a diagram to show the unit cell of ReO 3 . Explain the relationship between your...
-
What are the different relays that employed for protection of apparatus and transmission lines?
-
In 1998, Boston Beer produced more than two dozen styles of beer. Then a few years later it was down to just a few. Now its up to more than 21 again. Do you see any problems with this?
-
The C.D. Howe Institute Business Cycle Council is an arbiter of the chronologyof Canadian business cycles. Go to its website at https://www.cdhowe.org/council /business-cycle-council a. How many...
-
With reference to Exercise 11.9, test the null hypothesis \(\beta=0.40\) against the alternative hypothesis \(\beta>\) 0.40 at the 0.05 level of significance. Data From Exercise 11.9 11.9 Scientists...
-
Selected financial data of two competitors, Home Depot and Lowes, are presented here. (All dollars are in millions.) Suppose the data were taken from the 2014 financial statements of each company....
-
Hurst, Inc. sold its 8% bonds with a maturity value of $9,000,000 on August 1, 2019 for $8,838,000. At the time of the sale the bonds had 5 years until they reached maturity. Interest on the bonds is...
-
Consider the three types of cubic units cells. (a) Assuming that the spherical atoms or ions in a primitive cubic unit cell just touch along the cubes edges, calculate the percentage of occupied...
-
The solid-state structure of silicon is shown below. (a) Describe this crystal as pc, bcc, or fcc. (b) What type of holes are occupied in the lattice? (c) How many Si atoms are there per unit cell?...
-
Using the central bank balance sheet diagrams, evaluate how each of the following shocks affects a countrys ability to defend a fixed exchange rate. In the following answers, we assume the central...
-
Review the following facts for four separate companies. Identify the two companies that lost money during the year, explaining how you reached your conclusion for each. COMPANY A Ending retained...
-
As you study accounting, you will become increasingly familiar with a variety of generally accepted accounting principles. Already, you are beginning to appreciate some of the fundamental principles,...
-
This chapter introduces many important terms and concepts. Certain of these terms and concepts are noted in the following listing. Find quotes/descriptions that explain/contrast each of the matched...
-
Determine whether each of the following items is an: (a) Cash (b) Dividend to shareholders (c) Land (d) Accounts payable (e) Capital stock (f) Notes payable (g) Accounts receivable (h) Salaries (i)...
-
If interest rates decline, which would you rather be holding, long-term bonds or short-term bonds? Why? Which type of bond has the greater interest-rate risk?
-
You're at a party and another guest comments that one way to overcome many of the accounting scandals would be to eliminate choice from IFRS. Provide a response to the guest.
-
Which task is performed by a book-keeper? A. Analysing the trading results B. Entering transactions in the ledger C. Preparing year-end financial statements D. Providing information for...
-
The 8-mm-diameter bolt is made of an aluminum alloy. It fits through a magnesium sleeve that has an inner diameter of 12 mm and an outer diameter of 20 mm. If the original lengths of the bolt and...
-
An acetal polymer block is fixed to the rigid plates at its top and bottom surfaces. If the top plate displaces 2 mm horizontally when it is subjected to a horizontal force P = 2 kN, determine the...
-
The rigid bar is pinned at A and supported by two aluminum rods, each having a diameter of 1 in., a modulus of elasticity E al = 10(10 3 ) ksi, and yield stress of (Ï Y ) al = 40 ksi. If the bar...
-
What are the implications for learning and development: Explain strategic learning plan alignment to organizational goals that will drive strategy. Define the scope of learning: What is covered by...
-
Explain how technology is currently used to transform manual business processes and provide insights for various stakeholders. Be sure to include the name of your example organization and at least...
-
Discuss in detail how Activity based costing and Activity based management can be used by companies in gaining a competitive advantage. You should also identify and discuss in detail one US, publicly...

Study smarter with the SolutionInn App


