Uracil is a base found in RNA. Indicate sites in the molecule where hydrogen bonding is possible
Question:
Uracil is a base found in RNA. Indicate sites in the molecule where hydrogen bonding is possible or that are sites of Lewis basicity.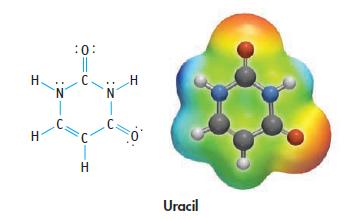
Transcribed Image Text:
H H N :0: || C N 1 C. H Uracil
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Uracil has two main functional groups that are relevant to hydrogen bonding and Lewis basic...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Proteins are synthesized with a particular amino acid sequence through the translation of information encoded in messenger RNA by an RNAprotein complex called a ribosome. Amino acids are specified by...
-
The RNA base uracil is a pyrimidine derivative, usually depicted in its keto form (page 396). The enol tautomer readily undergoes electrophilic substitution, unlike most pyrimidines. a. Draw the...
-
The operations manager of a company that produces frozen dinners had received numerous complaints from supermarkets about the companys Chick-n-Gravy dinners. The manager spilled/mixed items,...
-
Indicate what training methods you would use for each of the following jobs. Give reasons for your choices. a. File clerk b. Computer operator c. Automobile service station attendant d. Pizza maker...
-
Solve the systems in Problems 27-54 by solving the corresponding matrix equation with an inverse, if possible. \(\left\{\begin{array}{l}x+2 z=7 \\ 2 x+y=16 \\ -2 y+9 z=-3\end{array}ight.\)
-
Heritage, a general contractor, had filed a breach of contract action against an electrical subcontracting firm that had withdrawn its bid right before the contract was to be performed. A jury had...
-
Hassellhouf Companys trial balance at December 31, 2014, is presented below. All 2014 transactions have been recorded except for the items described shown below. Unrecorded transactions: 1. On May 1,...
-
Why did big business get bigger in the US than in other industrialized countries (e.g., Britain and Germany) during the 2d Industrial Revolution? How did the US differ from other countries such as...
-
Chemists often refer to the degree of ionization of a weak acid or base and give it the symbol . The equilibrium constant in terms of and Co, the initial acid or base concentration, is given by the...
-
You purchase a bottle of water. On checking its pH, you find that it is not neutral, as you might have expected. Instead, it is slightly acidic. Why?
-
What are one advantage and one disadvantage of the payback method?
-
Given this Gracenat Company income statement, answer a through e below. 2022 2021 2020 2019 Revenue 12,000 11,000 9,900 8,910 Cost of Goods Sold 6,900 6,353 5,693 5,159 Gross Profit 5,100 4,647 4,207...
-
Let ak be the infinite series 1+4+9+16+25+36 + ..., and let k=1 {n} be the corresponding sequence of partial sums. What are ag and 85 ? Your answers should be integers. ag = S5 81
-
1. A firm has beginning net fixed assets of $168,000 and ending net fixed assets of $141,300. Depreciation was $21,000. What is the amount of capital spending? A. $2,500 B. $5,700 C. $47,700 D,...
-
Assume today is July 1, 2023. A company has set a date to decide about the expansion or abandonment of a project at 6/30/2024. The Net Salvage Value at that date, if the company abandons the project,...
-
1. Two birds start ying from the tops of two towers 50 feet apart; one tower is 30 feet high and the other 40 feet high. Starting at the same time and ying at the same rate, the birds reach a...
-
Over the time covered here, did Hewlett-Packards board of directors fulfill its duties to the companys share owners? Explain how it met or did not meet basic duties.
-
SBS Company have received a contract to supply its product to a Health Care Service Hospital. The sales involve supplying 1,250 units every quarter, the sales price is RM 85 per unit. The Client...
-
The constant volume heat capacity for all monatomic gases is 12.48 J mol 1 K 1 . Why?
-
When are rotational degrees of freedom expected to contribute R or 3/2 R (linear and nonlinear, respectively) to the molar constant volume heat capacity? When will a vibrational degree of freedom...
-
The molar constant volume heat capacity of N 2 is 20.8 J mol 1 K 1 . What is this value in terms of R? Can you make sense of this value?
-
You are an analyst and have certain rates of return, profit margins, and other information on the Jeff, Matt, and Mike Associates Company. Using the information in the exhibit, complete the partial...
-
Each of the following independent companies is missing numerical data. Required: Use your knowledge of the financial statement equations and their interrelationships to fill in the missing amounts....
-
On 31 December 2016, the Summary Balance Sheet of John Brown Limited, booksellers, was as follows: Capital 10,000 Shop equipment 3,750 Creditors 5,000 Stock of books 6,000 Proposed dividends 750...

Study smarter with the SolutionInn App


