Using data in Appendix L, calculate r H, r G, and r S for
Question:
Using data in Appendix L, calculate ΔrH°, ΔrG°, and ΔrS° for the reaction of carbon and water to give CO and H2 at 298 K.
Data given in Appendix L
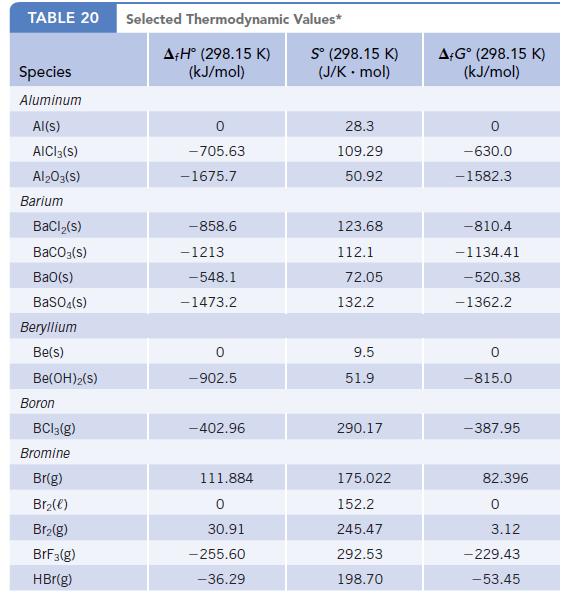
Transcribed Image Text:
TABLE 20 Species Aluminum Al(s) AICI 3(S) Al2O3(S) Barium BaCl₂(s) BaCO3(s) BaO(s) BaSO4(s) Beryllium Be(s) Be(OH)2(S) Boron BC13(g) Bromine Br(g) Br₂(e) Br₂(g) BrF3(g) HBr(g) Selected Thermodynamic A+Hº (298.15 K) (kJ/mol) 0 -705.63 -1675.7 -858.6 -1213 -548.1 -1473.2 -902.5 -402.96 111.884 0 30.91 -255.60 -36.29 Values* Sº (298.15 K) (J/K . mol) 28.3 109.29 50.92 123.68 112.1 72.05 132.2 9.5 51.9 290.17 175.022 152.2 245.47 292.53 198.70 A+Gᵒ (298.15 K) (kJ/mol) -630.0 -1582.3 -810.4 -1134.41 -520.38 -1362.2 0 -815.0 -387.95 82.396 0 3.12 - 229.43 -53.45
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
2 NO NO4 AH for NO22331662 KJmol AH for NO 908 KJmol AS for NO2 24004JmolK 28 AS ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Using data in Appendix C, calculate ÎH°, ÎS°, and ÎG° at 298 K for each of the following reactions. In each case show that ÎG° = ÎH° -...
-
In Section 11.5 we defined the vapor pressure of a liquid in terms of an equilibrium. (a) Write the equation representing the equilibrium between liquid water and water vapor and the corresponding...
-
The HaberBosch process for the production of ammonia is one of the key industrial processes in developed countries. N 2 (g) + 3 H 2 (g) 2 NH 3 (g) (a) Calculate r G for the reaction at 298 K, 800...
-
You are the senior auditor in charge of the December 31, 2018, year-end audit for Cleo Patrick Cosmetics Inc. (CPCI). CPCI is a large, privately held Canadian company that was founded in 1999 by one...
-
Restraining reliance before breach reduces the harm that it causes. Mitigating damages after breach also reduces the harm that it causes. In the Waffle Shop case, for example, Yvonne can mitigate...
-
What is 5ED4 - 07A4 when these values represent unsigned 16-bit hexadecimal numbers? The result should be written in hexadecimal. Show your work.
-
An OEM assembled drive assemblies for its pumps. The following photograph was taken on one of the manufactured drive cabinets. The power cable between the motor and the drive was grounded by...
-
Sentry Transport, Inc., of Atlanta provides in-town parcel delivery services in addition to a full range of passenger services. Sentry engaged in the following activities during the current year: a....
-
Required information Use the following information for the Exercises below. ( Algo ) [ The following information applies to the questions displayed below. ] Laker Company reported the following...
-
A method recently suggested for the preparation of hydrogen (and oxygen) from water proceeds as follows: (a) Sulfuric acid and hydrogen iodide are formed from sulfur dioxide, water, and iodine. (b)...
-
Write an equation for the reaction of potassium and hydrogen. Name the product. Is it ionic or covalent? Predict one physical property and one chemical property of this compound.
-
Franklin, John, Henry, and Harry have decided to pool their financial resources and business skills in order to open up and run a coffee shop. They will share any profits or losses that the business...
-
Lets say you work in Illinois, and you want to figure out how much income tax you need to pay for the year. Here is what you know: Your hourly wage is $10. You have worked 1000 hours for the year ...
-
Ahson Tariq is director of operations for CTC. CTC specializes in global merchandising of the world's finest cotton fibers. It is common practice for CTC to purchase cotton in bulk from regional...
-
Lets create a new program where we determine if a person needs to file taxes or not based on age and the income they make. Create a new class file called TaxIndicator and create a main() container....
-
The op-amp circuit shown in Figure 6.80 is an active low-pass filter. Derive the input-output differential equation relating the output voltage \(v_{0}(t)\) and the input voltage...
-
This problem requires you to select three stocks for a hypothetical trading securities investment portfolio. Assume you invest $100,000 in each company at beginning of "day 1." Prepare a daily...
-
Robert Kapela owned and managed a franchise of Ithaca Espresso, Incorporated. The company's balance sheet accounts had the following balances on September 1, 20X0, the beginning of a fiscal year:...
-
Arlington Merchants reported the following on its income statement for the fiscal years ending December 31, 2016 and 2015. 2016 2015 Sales $4,857,500 $4,752,900 Cost of goods sold 3,258,950 3,207,000...
-
Make a hypothetical sketch of a velocitytime graph in which the velocity is always positive, but the acceleration is always negative. Give a physical example of such motion.
-
An object is found to move with an acceleration of magnitude 12 m/s 2 when it is subjected to a force of magnitude 200 N. Find the mass of the object.
-
Write the expression for computing the pressure in a fluid.
-
Maben Company was started on January 1, Year 1, and experienced the following events during its first year of operation: 1. Acquired $30,000 cash from the issue of common stock. 2. Borrowed $40,000...
-
The Devon Motor Company produces automobiles. On April 1st the company had no beginning inventories and it purchased 6,290 batteries at a cost of $95 per battery. It withdrew 5,800 batteries from the...
-
Maben Company was started on January 1, Year 1, and experienced the following events during its first year of operation: 1. Acquired $30,000 cash from the issue of common stock. 2. Borrowed $40,000...

Study smarter with the SolutionInn App


