Using Figure 17.11, suggest an indicator to use in each of the following titrations. (a) NaHCO 3
Question:
Using Figure 17.11, suggest an indicator to use in each of the following titrations.
(a) NaHCO3 is titrated to CO32− with NaOH.
(b) Hypochlorous acid is titrated with NaOH.
(c) Trimethylamine is titrated with HCl.
Data given in Figure 17.11
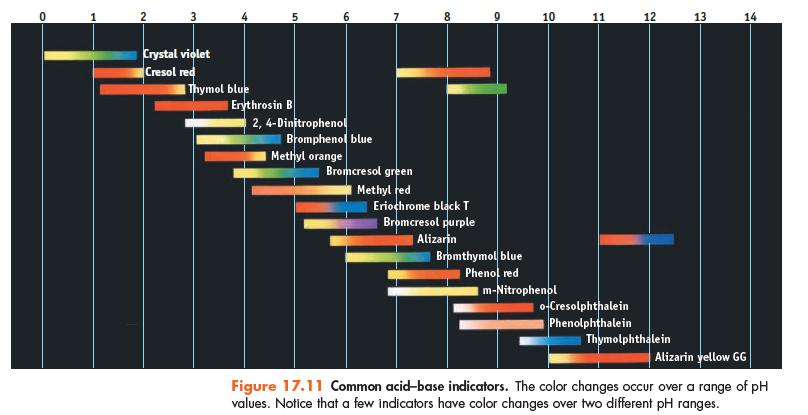
Transcribed Image Text:
3 Crystal violet Cresol red Thymol blue 5 Erythrosin B 2,4-Dinitrophenol Bromphenol blue Methyl orange 7 Bromcresol green Methyl red 00 Eriochrome black T Bromcresol purple Alizarin 8 10 Bromthymol blue Phenol red Im-Nitrophenol 11 o-Cresolphthalein Phenolphthalein 12 Thymolphthalein 13 14 Alizarin yellow GG Figure 17.11 Common acid-base indicators. The color changes occur over a range of pH values. Notice that a few indicators have color changes over two different pH ranges.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a NaHCO3 is titrated to CO32 with NaOH Phenolphthalein indic...View the full answer

Answered By

Jonas Araujo
I have recently received the degree of PhD. In Physics by the Universidade Federal do Maranhão after spending a term in Durham University, as I have been awarded a scholarship from a Brazilian mobility program. During my PhD. I have performed research mainly in Theoretical Physics and published works in distinguished Journals (check my ORCID: https://orcid.org/0000-0002-4324-1184).
During my BSc. I have been awarded a scholarship to study for a year in the University of Evansville, where I have worked in detection-analysis of photon correlations in the the Photonics Laboratory. There I was a tutor in Electromagnetism, Classical Mechanics and Calculus for most of that year (2012).
I am very dedicated, honest and a fast learner, but most of all, I value a job well done.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Using Figure 17.11, suggest an indicator to use in each of the following titrations: (a) The weak base pyridine is titrated with HCl. (b) Formic acid is titrated with NaOH. (c) Ethylenediamine, a...
-
Suggest a suitable indicator for a titration based on each of the following reactions. Use 0.05 M if an equivalence point concentration is needed. *(a) H2CO3 + NaOH NaHCO3 + H2O (b) H2P + 2NaOH ...
-
Del Gato Clinic's cash account shows an $14,180 debit balance and its bank statement shows $13,236 on deposit at the close of business on June 30. a. Outstanding checks as of June 30 total $1,502. b....
-
Carl contributes equipment with a $50,000 adjusted basis and an $80,000 FMV to Cook Corporation for 50 of its 100 shares of stock. His son, Carl Jr., contributes $20,000 cash for the remaining 50...
-
What are some reasons for the implementation of ESOPs? Cite some of the potential problems concerning their use.
-
Let X 1 , . . . , X n be an i.i.d. Bernoulli sequence with parameter p. (a) Find the conditional distribution of X 1 given X 1 + + X n = k. (b) Find E[X 1 |X 1 + + X n = k] and V[X 1 |X 1 + ...
-
Determined to reduce the \(\$ 7\) per week cost associated with heat loss through their patio window by convection and radiation, the tenants of Problem 9.15 cover the inside of the window with a...
-
George Parker was paid a salary of $74,700 during 2013 by Umberger Company. In addition, during the year, Parker started his own business as a public accountant and reported a net business income of...
-
A discussion of THREE separate ESG issues from Air canada company's report, one each addressing Environmental (CHAR), Social and Governance issues. Evaluate how each issue relates to the disclosure...
-
Name two insoluble salts of each of the following ions. (a) Cl (b) Zn 2+ (c) Fe 2+
-
Construct a rough plot of pH versus volume of base for the titration of 25.0 mL of 0.050 M HCN with 0.075 M NaOH. (a) What is the pH before any NaOH is added? (b) What is the pH at the halfway point...
-
Solve the given system of equations using either Gaussian or Gauss-Jordan elimination. V2x + y + V2y -y + V2z 2z = 3z = -V2
-
Compare and contrast positive and negative externalities.
-
Discuss how the term market failure is understood in the context of public financial management.
-
Discuss the sources of revenue for national government.
-
Compare and contrast conditional and unconditional grants
-
Briefly summarise the key aspects of GAAP, GRAP and GAMAP as accounting standards.
-
Crismon Company makes camping tents in a single production department. All direct materials are added at the beginning of the manufacturing process. Information for the month of July follows:...
-
Define a traverse in Surveying?
-
A crude model for the molecular distribution of atmospheric gases above the Earths surface (denoted by height, h) can be obtained by considering the potential energy due to gravity: In this...
-
Another use of distribution functions is determining the most-probable value, which is done by realizing that at the distribution maximum the derivative of the distribution function with respect to...
-
In nonlinear optical switching devices based on dye-doped polymer systems, the spatial orientation of the dye molecules in the polymer is an important parameter. These devices are generally...
-
Done 3:20 5G Assignments > Assignment 1 - Chapter 3 (Flowchart) Assignment 1 - Chapter 3 (Flowchart) Hide Assignment Information Instructions Please complete a document flowchart in Excel that...
-
1. Determine whether the argument given is valid or invalid. That... 1. Determine whether the argument given is valid or invalid. That is, does the conclusion logically follow from the given true...
-
Please help me to answer these questions Thank you this is... Please help me to answer these questions Thank you this is table Table 11-1: https://www.webassign.net/brecmbc9/11-table-1.pdf 1. This...

Study smarter with the SolutionInn App


