Write a balanced chemical equation for the preparation of H 2 (and CO) by the reaction of
Question:
Write a balanced chemical equation for the preparation of H2 (and CO) by the reaction of CH4 and water. Using data in Appendix L, calculate ΔrH°, ΔrG°, and ΔrS° for this reaction at 298 K.
Data given in Appendix L
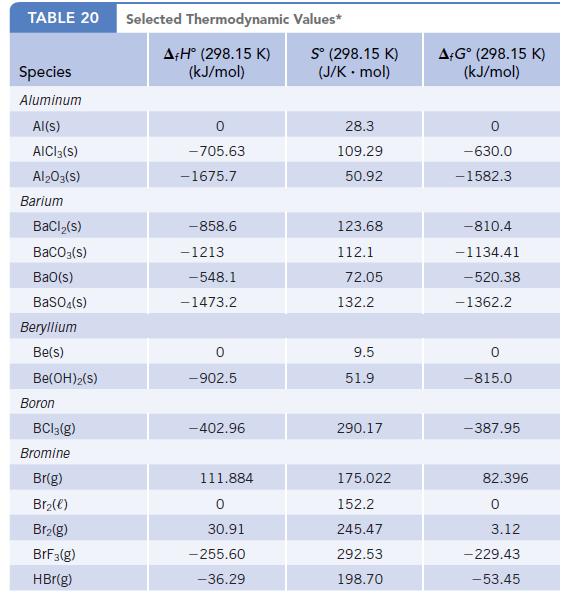
Transcribed Image Text:
TABLE 20 Species Aluminum Al(s) AICI 3(S) Al2O3(S) Barium BaCl₂(s) BaCO3(s) BaO(s) BaSO4(s) Beryllium Be(s) Be(OH)2(S) Boron BC13(g) Bromine Br(g) Br₂(e) Br₂(g) BrF3(g) HBr(g) Selected Thermodynamic A+Hº (298.15 K) (kJ/mol) 0 -705.63 -1675.7 -858.6 -1213 -548.1 -1473.2 -902.5 -402.96 111.884 0 30.91 -255.60 -36.29 Values* Sº (298.15 K) (J/K . mol) 28.3 109.29 50.92 123.68 112.1 72.05 132.2 9.5 51.9 290.17 175.022 152.2 245.47 292.53 198.70 A+Gᵒ (298.15 K) (kJ/mol) -630.0 -1582.3 -810.4 -1134.41 -520.38 -1362.2 0 -815.0 -387.95 82.396 0 3.12 - 229.43 -53.45
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
The balanced chemical equation for the preparation of H2 and CO by the r...View the full answer

Answered By

Douglas Makokha
Unlock Academic Success with Dedicated Tutoring and Expert Writing Support!
Are you ready to excel in your academics? Look no further! As a passionate tutor, I believe that dedication and hard work are the keys to achieving outstanding results. When it comes to academics, I strive to provide nothing but the best for every student I encounter.
With a relentless thirst for knowledge, I have extensively researched numerous subjects and topics, equipping myself with a treasure trove of answers to tackle any question that comes my way. With four years of invaluable experience, I have mastered the art of unraveling even the most intricate problems. Collaborating with esteemed writers has granted me exclusive access to the trade secrets utilized by the industry's top professionals.
Allow me the pleasure of assisting you with your writing assignments. I thrive on challenges and will guide you through any obstacles you may face. Together, we will unlock your academic potential and pave the way for your success.
4.90+
60+ Reviews
342+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Write a balanced chemical equation for the pentose phosphate pathway in the first two modes depicted in Figure 12.36, where (a) ribose-5-phosphate synthesis is maximized (b) NADPH production is...
-
A historian discovers a nineteenth-century notebook in which some observations, dated 1822, were recorded on a substance thought to be a new element. Here are some of the data recorded in the...
-
Write a balanced chemical equation for the reaction that occurs when (a) Calcium metal undergoes a combination reaction with O2(g) (b) Copper(II) hydroxide decomposes into copper(II) oxide and water...
-
Kamloops Company is a grocery wholesaler and is planning to expand its operations. The company has asked the bank for a loan to finance the expansion. Alphonzo, the companys manager, has prepared the...
-
State the Coase Theorem as applied to remedies for breach of contract.
-
A cue ball bounces off the rail of a pool table as shown in Figure Q7.12. The momentum of the ball changes.? (a) What is the source of the impulse?? (b) That is, what is the source of the force?? (c)...
-
Name the measurement tool seen in the following photograph. www
-
Oceanic Airways makes investments in available-for-sale securities. Selected income statement items for the years ended December 31, 2012 and 2013, plus selected items from comparative balance...
-
On December 3 1 , 2 0 2 4 , Sheridan Landscaping's common shares were trading for $ 1 5 , and the company had the following shareholder's equity accounts:Common Shares ( no par value, unlimited...
-
Write an equation for the reaction of potassium and hydrogen. Name the product. Is it ionic or covalent? Predict one physical property and one chemical property of this compound.
-
Write balanced chemical equations for the reaction of hydrogen gas with oxygen, chlorine, and nitrogen.
-
According to the pure expectations theory of interest rates, how much do you expect to pay for a five-year STRIPS on February 15, 2009? How much do you expect to pay for a two-year STRIPS on February...
-
A function \(f(x)\) is said to be concave over an interval \((a, b)\) if it satisfies the property \[ f\left\{\lambda x_{1}+(1-\lambda) x_{2} ight\} \geq \lambda f\left(x_{1} ight)+(1-\lambda)...
-
Design a class named Car that has the following fields: Next, design a program that creates a Car object, and then calls the accelerate method five times. After each call to the accelerate method,...
-
A diet center claims that people subscribing to its program lose an average of 4 lb in the first week of the diet. Suppose 25 people in the diet centers program are chosen at random and are found to...
-
Consider two risky assets with returns \(\tilde{r}_{1}, \tilde{r}_{2}\), with corresponding expected returns \(e_{1}, e_{2}\), variances \(\sigma_{1}^{2}, \sigma_{2}^{2}\) and correlation \(ho\). Let...
-
Design an Essay class that extends the GradedActivity class presented in this chapter. The Essay class should determine the grade a student receives for an essay. The students essay score can be up...
-
Refer to problem 3-37. Prepare a balance sheet as of November 30, 20X0, and an income statement for the month of November. Prepare the retained earnings column of a statement of stockholders' equity....
-
Following is the current balance sheet for a local partnership of doctors: The following questions represent independent situations: a. E is going to invest enough money in this partnership to...
-
Write the expression relating gage pressure, absolute pressure, and atmospheric pressure.
-
The value for the absolute pressure will always be greater than that for the gage pressure. Are (or can be) true or false. For those that are false, explain why.
-
As long as you stay on the surface of Earth, the atmospheric pressure will be 14.7 psia. Are (or can be) true or false. For those that are false, explain why.
-
During the month, Bavarian Auto Co. received $476,500 in cash and paid out $402,150 in cash. a. Does this information indicate that Bavarian Auto Co. had net income of $74,350 during the month? Net...
-
Cash Accounts Receivable Inventory Property Plant & Equipment Other Assets Total Assets 16,800 Total Liabilities 9,900 Accounts Payable 2,700 4,500 Debt 3,500 3,800 Other Liabilities 1,000 7,200...
-
The condensed income statement for the Consumer Products Division of Hydra Industries Inc. is as follows (assuming no support department allocations): Sales $1,908,000 Cost of goods sold (858,600)...
Multiple Testing Procedures With Applications To Genomics 1st Edition - ISBN: 1441923799 - Free Book

Study smarter with the SolutionInn App


