Question: As the voltaic cell shown here runs, the blue solution gradually gets lighter in color and the gray solution gets darker. (a) What species is
As the voltaic cell shown here runs, the blue solution gradually gets lighter in color and the gray solution gets darker.
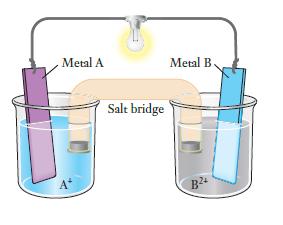
(a) What species is oxidized and what is reduced?
(b) Which electrode is the anode and which is the cathode?
(c) Which metal electrode gains mass?
(d) In which direction do electrons flow through the external circuit?
Metal A Salt bridge Metal B. B24
Step by Step Solution
★★★★★
3.39 Rating (158 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

a B is oxidized and A is ... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock


