Question: The compound shown below forms an amorphous solid (a glass) at room temperature and has been used as a medium for storing information holographically. Write
The compound shown below forms an amorphous solid (a glass) at room temperature and has been used as a medium for storing information holographically. Write the correct molecular formula for this molecule.
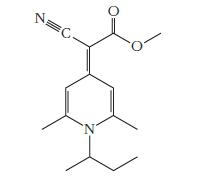
N C. N
Step by Step Solution
3.49 Rating (156 Votes )
There are 3 Steps involved in it
The compound shown in the image is triphenylphosphine oxide which has the molecular formula C6H5POO2 ... View full answer

Get step-by-step solutions from verified subject matter experts


