Which of the following reactions is (are) spontaneous at standard conditions? 3+ 2+ (a) Zn(s) + 2
Question:
Which of the following reactions is (are) spontaneous at standard conditions?
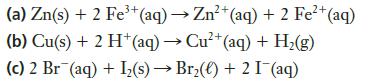
Transcribed Image Text:
3+ 2+ (a) Zn(s) + 2 Fe³+ (aq) → Zn²+ (aq) + 2 Fe²+ (aq) (b) Cu(s) + 2 H+ (aq) → Cu²+ (aq) + H₂(g) (c) 2 Br (aq) + I₂(s)→ Br₂() +2 I¯(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The reaction a Zns 2 Fe3aq Znaq 2 Feaq is not spontaneous at standard conditions ...View the full answer

Answered By

Gauri Hendre
I worked as EI educator for Eduphy India YT channel. I gave online tutorials to the students who were living in the villages and wanted to study much more and were preparing for NEET, TET. I gave tutions for topics in Biotechnology. I am currently working as a tutor on course hero for the biochemistry, microbiology, biology, cell biology, genetics subjects. I worked as a project intern in BAIF where did analysis on diseases mainly genetic disorders in the bovine. I worked as a trainee in serum institute of India and Vasantdada sugar institute. I am working as a writer on Quora partner program from 2019. I writing on the topics on social health issues including current COVID-19 pandemic, different concepts in science discipline. I learned foreign languages such as german and french upto A1 level. I attended different conferences in the science discipline and did trainings in cognitive skills and personality development skills from Lila Poonawalla foundation. I have been the member of Lila poonawalla foundation since 2017. Even I acquired the skills like Excel spreadsheet, MS Office, MS Powerpoint and Data entry.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
Which of the following reactions are spontaneous in the direction written? See Table 18.2 for data. a. C(graphite) + 2H2(g) CH4(g) b. 2H2(g) + O2(g) 2H2O(l ) c. 4HCN(g) + 5O2(g) 2H2O(l) + 4CO2(g)...
-
A disproportionation reaction involves a substance that acts as both an oxidizing agent and a reducing agent, producing higher and lower oxidation states of the same element in the products. Which of...
-
From the values of H and S, predict which of the following reactions would be spontaneous at 25C: Reaction A: H 5 10.5 kJ/mol, DS 5 30 J/K ? mol; reaction B: H 5 1.8 kJ/mol, S 5 2113 J/K ? mol. If...
-
Use the ideas of Richardson's extrapolation and Romberg's method to evaluate the first derivative of f = ex at x = 0. To construct the first column of the Romberg's table, use the formula (exth -...
-
What could be included in an effective opening and closing of a claim or complaint letter?
-
Journalize, record, and post when appropriate the following transactions into the general journal (p. 2) for Kadens Clothing. All purchases discounts are 4/10, n/30. If using working papers, be sure...
-
Find f0.01 for F12,8.
-
Normal Corporation uses standard costing and is in the process of updating its direct materials and direct labor standards for Product 20B. The following data have been accumulated: Direct materials...
-
1) Explain how the process of valuing a high-growth company differs from valuing an established company. 2) How does the total market for a new product differ from a company's addressable market?...
-
Consult a table of standard reduction potentials and determine which of the following reactions are spontaneous under standard electrochemical conditions. 2+ (a) Mn(s) + 2 H+ (aq) H(g) + Mn+ (aq)...
-
Suppose that you cannot find a table of standard reduction potentials. You remember that the standard reduction potential of Cu 2+ + 2 e Cu(s) is 0.337 V. Given that Gf(Cu 2+ ) = 65.49 kJ mol 1 and...
-
Keeler et al. (2004) estimate that the U.S. Tobacco Settlement between major tobacco companies and 46 states caused the price of cigarettes to jump by 45 per pack (21%) and overall per capita...
-
lan operates an automobile repair shop and he has the opportunity to buy an additional machine, which he needs to continue servicing his increasing customer base, at a bargain price of $6,200 if he...
-
The Bronco Corporation exchanged land for equipment. The land had a book value of $129,000 and a fair value of $168,000. Bronco paid the owner of the equipment $19,000 to complete the exchange which...
-
On December 31, 2024, TB Inc. borrowed $1,800,000 at 11% payable annually to finance the construction of a new building. In 2025, the company made the following expenditures related to this building:...
-
Information from the financial statements of ABC Corporation included the following at December 3 1 , 2 0 2 1 : Common shares outstanding throughout the year 1 0 0...
-
A company is considering the purchase of equipment that costs $ 1 2 0 , 0 0 0 and has an estimated residual value of $ 1 0 , 0 0 0 after its 5 year useful life. It is expected to generate total net...
-
Carleton Builders Ltd. recorded the following summarized transactions during the current year: a. The company originally sold and issued 100,000 common shares. During the current year, 94,000 of...
-
Write the statement to store the contents of the txtAge control in an Integer variable named intAge.
-
The solid shaft of radius c is subjected to a torque T at its ends. Show that the maximum shear strain in the shaft is γ max = T c /JG. What is the shear strain on an element located at...
-
The propellers of a ship are connected to an A-36 steel shaft that is 60 m long and has an outer diameter of 340 mm and inner diameter of 260 mm. If the power output is 4.5 MW when the shaft rotates...
-
A motor delivers 500 hp to the shaft, which is tubular and has an outer diameter of 2 in. If it is rotating at 200 rad/s, determine its largest inner diameter to the nearest 1/8 in. if the allowable...
-
Penne Pharmaceuticals sold 15 million shares of its $1 par common stock to provide funds for research and development. If the issue price is $12 per share, what is the journal entry to record the...
-
Case Defence sells smartphone cases and uses the perpetual inventory system. The following is information on the purchases and sales of "Defender Box" cases. On October 1, Case Defence had 40 units...
-
Following is information on an investment in a manufacturing machine. The machine has zero salvage value. The company requires a 6% return from its investments. Initial investment Net cash flows:...

Study smarter with the SolutionInn App


