Following is information on an investment in a manufacturing machine. The machine has zero salvage value....
Fantastic news! We've Found the answer you've been seeking!
Question:
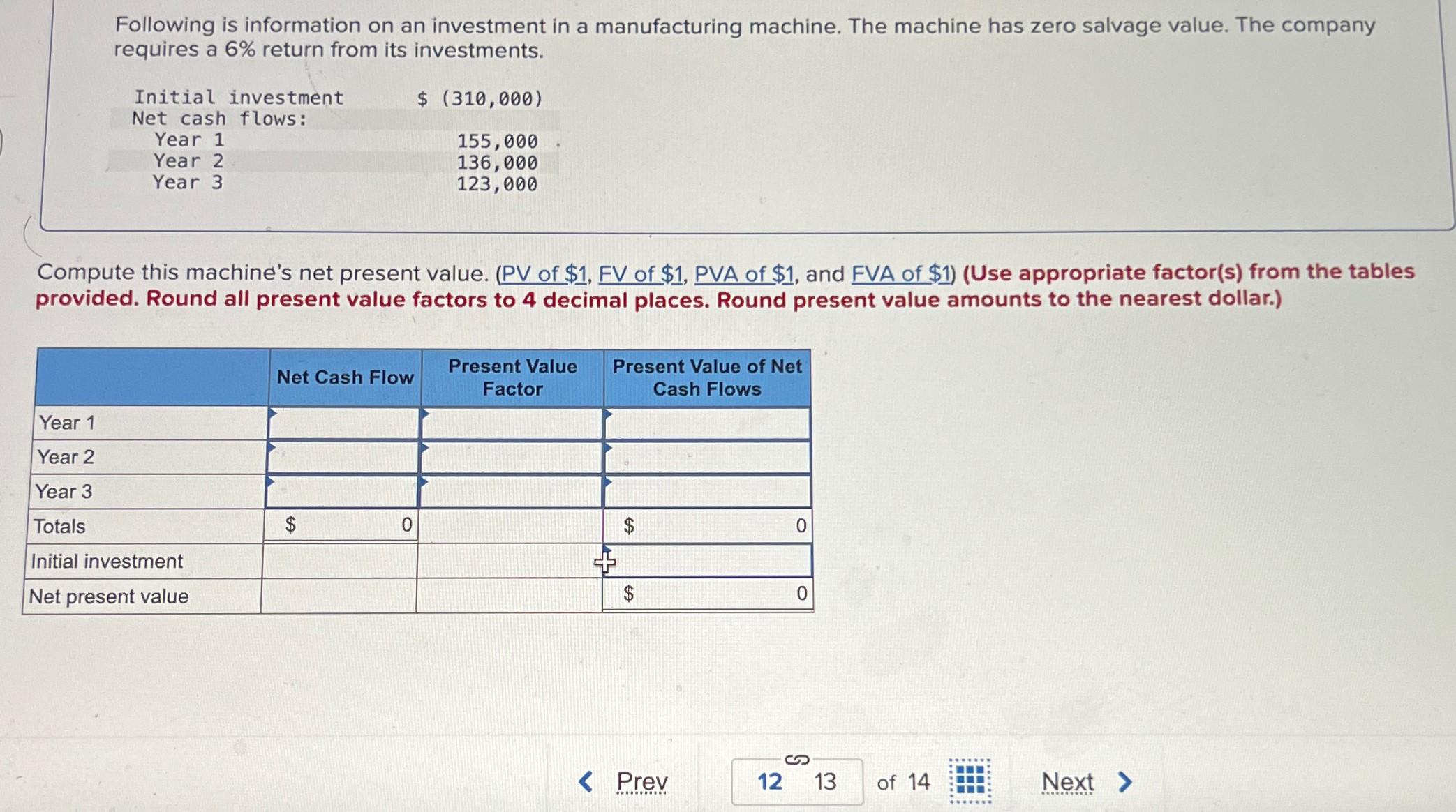
Transcribed Image Text:
Following is information on an investment in a manufacturing machine. The machine has zero salvage value. The company requires a 6% return from its investments. Initial investment Net cash flows: Year 1 Year 2 Year 3 $ (310,000) 155,000 136,000 123,000 Compute this machine's net present value. (PV of $1, FV of $1, PVA of $1, and FVA of $1) (Use appropriate factor(s) from the tables provided. Round all present value factors to 4 decimal places. Round present value amounts to the nearest dollar.) Year 1 Year 2 Year 3 Totals Initial investment Net present value Net Cash Flow Present Value Factor Present Value of Net Cash Flows EA $ 0 $ 0 $ 0 S Prev 12 13 of 14 Next > Following is information on an investment in a manufacturing machine. The machine has zero salvage value. The company requires a 6% return from its investments. Initial investment Net cash flows: Year 1 Year 2 Year 3 $ (310,000) 155,000 136,000 123,000 Compute this machine's net present value. (PV of $1, FV of $1, PVA of $1, and FVA of $1) (Use appropriate factor(s) from the tables provided. Round all present value factors to 4 decimal places. Round present value amounts to the nearest dollar.) Year 1 Year 2 Year 3 Totals Initial investment Net present value Net Cash Flow Present Value Factor Present Value of Net Cash Flows EA $ 0 $ 0 $ 0 S Prev 12 13 of 14 Next >
Expert Answer:

Posted Date:
Students also viewed these accounting questions
-
Project management is a systematic approach to defining, planning, executing, and closing projects to achieve specific goals and objectives. By addressing specific questions related to the...
-
On average, do people prefer a... 6 game package with 300-level, mid-court seats and a $20 gift certificate for $35/seat or a 3 game package with 300-level, corner seats that come with a hotdog and...
-
Go to www.sec.gov to research how a new security is approved by the Securities and Exchange Commission. What is the process involved and how long would it take? Next, contact a financial institution...
-
Two resistors connected in series have an equivalent resistance of 690 . When they are connected in parallel, their equivalent resistance is 150 . Find the resistance of each resistor.
-
Sherry Taft and I.aura Sims are opening Bank Compliance Consultants. The area is growing, and no competitors are located nearby. Their basic decision is how to organize the business. Taft thinks the...
-
At an initiation banquet for new members of your business club, you are talking with two college students who are majoring in marketing. In discussing the accounting course they are taking, they...
-
Lori is a new hire and is keen to show her prowess in delta hedging to her boss. She reviews the put options on Waste Connections Ltd. and sells 60,000 put options. Spot price on the date of trading...
-
Option e asks us to investigate how the cycle efficiency changes with the value of q, and what parameter we could change to increase or decrease q. In a steam power plant, the thermal efficiency is...
-
What are the two types of files that auditors review in preparation for an audit? What are the contents of each file?
-
What are the five objectives of internal control systems?
-
How does the Standards describe the scope (in general terms) of an internal audit?
-
Outline the key aspects of the preliminary communication for an audit.
-
What three types of audit approval are given for audits? Discuss each briefly.
-
1. Write a program to solve the 8-puzzle problem using the uninformed searching techniques breadth-first search (BFS) and iterative deepening search (IDS). The input to your program are the numbers...
-
What is a make-or-buy decision?
-
Mention the areas of application of the fuel cell.
-
(a) Describe the effect of temperature on the equilibrium constant. (b) Industrial \(\mathrm{CH}_{3} \mathrm{OH}\) is prepared according to the reaction \[ \mathrm{CO}(\mathrm{g})+2...
-
Estimate the standard free energy change and equilibrium constant at \(700 \mathrm{~K}\) for the reaction \[ \mathrm{N}_{2}(\mathrm{~g})+3 \mathrm{H}_{2}(\mathrm{~g})=2 \mathrm{NH}_{3}(\mathrm{~g})...

Study smarter with the SolutionInn App

