A mixture of sulfur dioxide and oxygen gas reacts as shown below. (a) Write the balanced
Question:
A mixture of sulfur dioxide and oxygen gas reacts as shown below.
(a) Write the balanced equation (remember to express the coefficients as the lowest set of whole numbers).
(b) Name the product.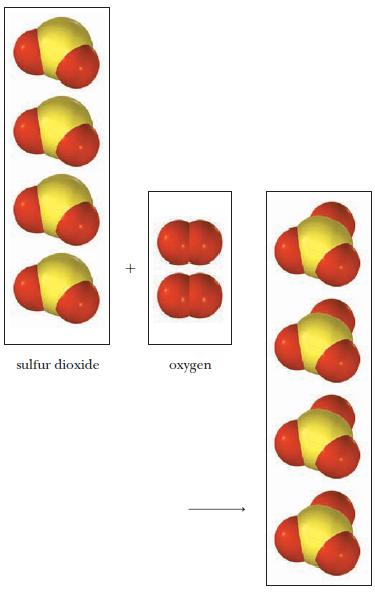
Transcribed Image Text:
sulfur dioxide oxygen
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Completed and balanced c...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
A mixture of carbon monoxide and oxygen gas reacts as shown below. (a) Write the balanced equation (remember to express the coefficients as the lowest set of whole numbers). (b) Name the product....
-
Iron pyrite, FeS 2 , is the form in which much of the sulfur exists in coal. In the combustion of coal, oxygen reacts with iron pyrite to produce iron(III) oxide and sulfur dioxide, which is a major...
-
Sulfuric acid is manufactured by the Contact process, in which a mixture of sulfur dioxide and excess air, preheated to 450C, is passed over a catalyst. a. How is the sulfur dioxide made? Give an...
-
discuss case study a bout remote analysis during covid 1 9 - 1 9 virus
-
Find the input impedance of a short-circuited coaxial transmission line of figure if Z o = 65 + j38?, ? = 0.7 + j2.5 /m, ? = 0.8 m. 0
-
a) Find and simplify the equation of the curve 3 + f(x 1). b) Describe the transformation. f(x) = x(x + 3)
-
What are the differences between average hourly earnings and the employment cost index?
-
Legislative Act: Section 41-1-6-9 of the state statutes defines defamation as the intentional publication of a false statement about a person. The statute defines publication as communication to a...
-
4. The atomic numbers of elements, U, V, W and X are 13, 14, 16 and 17 respectively. Which one of the elements will react with oxygen to form an ionic compound? (Atomic number of oxygen is 8) X A. B....
-
This problem continues the process of preparing the Kipley Company's Employee Payroll Register for the pay period ending January 8th, 20--. In previous chapters, gross wages were computed for each...
-
Write balanced equations for the following reactions. (a) C5H12 + O CO + HO (b) NH3 + O N + HO (c) KOH + HSO4 KSO4 + HO
-
Given the following equation, calculate the mass (in grams) of AlCl 3 that can be produced from 4.40 g Al and 12.0 g Cl 2 ? 2A1+3C1 2A1C13
-
Given the information below, compute annualized returns: Price Initial Price Time Period 15 months 11 months Income Asset Change $ 2 $ 6 10 $29 40 B 70 30 7 years 24 months 50 20 -8 D
-
A U.S. publisher sells $200 worth of books to a resident of Brazil. By itself, this item increases the U.S. current account balance. Describe some offsetting transactions that could ensure that the...
-
When liquid water freezes, is heat released to the surroundings or absorbed from the surroundings?
-
Which has more atomsa 1-gram sample of carbon-12 or a 1-gram sample of carbon-13? Explain.
-
Why is perfume typically applied behind the ear rather than on the ear?
-
Two-tailed test with \(\alpha=0.10\), degrees of freedom in the numerator \(=12\), degrees of freedom in the denominator \(=20\). Find the critical value( \(s\) ) for \(\alpha\).
-
You are the newest member of the staff of Brinks & Company, a medium-size investment management firm. Les Kramer, an employee of two years, is your supervisor. Les has a reputation as being...
-
Organizations are increasing their use of personality tests to screen job applicants. What are some of the advantages and disadvantages of this approach? What can managers do to avoid some of the...
-
Predict the major product obtained when each of the following compounds is treated with fuming sulfuric acid: (a) Chlorobenzene (b) Phenol (c) Benzaldehyde (d) Ortho-Nitrophenol (e) Para-Bromotoluene...
-
For each of the following groups, identify whether it is an activator or a deactivator, and determine its directing effects: (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) -OMe
-
Starting with benzene and using any other necessary reagents of your choice, design a synthesis for each of the following compounds: (a) (b) (c) Br-
-
1. In an isolated calorimeter, 105 g of water is placed in a 20-g aluminum cup. Both the water and the cup start at 32 C. 15 g of ice, initially at 0 C is added to the water and the system is allowed...
-
Self-Travel, a car rental firm, has two methods of charging for car rental: Method 1 $64 per day + 25 cents per kilometre Method 2 $89 per day with unlimited travel a Write a rule for each method if...
-
18. Lets pretend that there is no air resistance. Determine the potential and kinetic energy for a 1 kg ball dropped at a height of 200 meters at for 3 seconds. SHOW ALL WORK FOR CREDIT. Time of Ball...

Study smarter with the SolutionInn App


