At 3000 K, carbon dioxide dissociates: CO(g) CO(g) + + 1/1/0(g) If Kp for this reaction is
Question:
At 3000 K, carbon dioxide dissociates: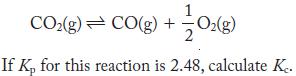
Transcribed Image Text:
CO(g) CO(g) + + 1/1/0(g) If Kp for this reaction is 2.48, calculate Ke.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The given chemical equation for the dissociation of carbon dioxide is CO...View the full answer

Answered By

Diane Joyce Pastorin
Please accept my enthusiastic application to solutioninn. I would love the opportunity to be a hardworking, passionate member of your tutoring program. As soon as I read the description of the program, I knew I was a well-qualified candidate for the position.
I have extensive tutoring experience in a variety of fields. I have tutored in English as well as Calculus. I have helped students learn to analyze literature, write essays, understand historical events, and graph parabolas. Your program requires that tutors be able to assist students in multiple subjects, and my experience would allow me to do just that.
You also state in your job posting that you require tutors that can work with students of all ages. As a summer camp counselor, I have experience working with preschool and kindergarten-age students. I have also tutored middle school students in reading, as well as college and high school students. Through these tutoring and counseling positions, I have learned how to best teach each age group.
4.60+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
One mole of water and one mole of CO2 are placed in a reactor at 3000 K and a total pressure of 400 kPa. The water dissociates into hydrogen and oxygen, and the carbon dioxide dissociates into carbon...
-
Consider the equilibrium dissociation of carbon dioxide CO 2 CO + 1/2 O 2 . At 2500 K, the equilibrium constant is 0.03635. Calculate the enthalpy of reaction for this reaction at 2500 K and use...
-
Carbon dioxide gas enters a pipe at 3 MPa and 500 K at a rate of 2 kg/s. CO2 is cooled at constant pressure as it flows in the pipe and the temperature CO2 drops to 450 K at the exit. Determine the...
-
The mass-to-charge ratio for the positive ion F+ is 1.97 107 kg/C. Using the value of 1.602 1019 C for the charge on the ion, calculate the mass of the fluorine atom. (The mass of the electron is...
-
Lets be more practical. DCF is not the gospel. Managers should not become so enchanted with DCF that strategic considerations are overlooked. Do you agree? Explain.
-
21st Century Insurance offers mail-order automobile insurance to preferred risk drivers in the Los Angeles area. The company is the low-cost provider of insurance in this market but doesn't believe...
-
When should we use packaging? Discuss its pros and cons.
-
During 2015, LeBron Corporation accepts the following notes receivable. a. On April 1, LeBron provides services to a customer on account. The customer signs a four-month, 9% note for $7,000. b. On...
-
Greg Quigley earned $5489 last year. There is no SUTA tax rate where he lives. How much FUTA tax must Greg's company pay for him if FUTA tax is currently 6.2% of the first $7000 earned by the...
-
Sulfur dioxide reacts with chlorine when sealed in a reactor at increased temperature. At 227 C, K c = 20.9. Calculate K p at this same temperature. SO(g) + Cl(g) SOCl(g)
-
Nitrosyl bromide is formed from nitrogen monoxide and bromine: K p for this reaction is 116 at 25 C. Calculate K c at this temperature. 1 NO(g) + - Br(g) NOBr(g) 2
-
Net sales, net income, and common shareholders' equity for Naturah Products Ltd. for a six-year period follow: Required 1. Compute trend percentages for 2010 through 2014, using 2009 as the base...
-
Standard Bertrand and Cournot competition assumes simultaneous move. Suppose now that, because of size or industry norms, one firm is always the first to set price or output, with a second firm...
-
This activity requires you to view the world using the perspective of the social construction of disability. Before you begin, re-read the entire Wendell article. The task is to describe something...
-
What is your understanding of the concept of elasticity? Does it makes sense to you? Provide also at least one example drawn from your experience as an active market participant. In other words, make...
-
Scarcity is a problem only for poor countries. Economics is the study of how to use unlimited resources to satisfy limited wants. If Jack slept in instead of either going to school or jogging, the...
-
Choose a highly visible leader, someone you admire and would like to emulate. You will be looking to examine a situation when a leader's cultural adaptation, cultural intelligence, ethics, and...
-
Suppose that JPMorgan Chase sells call options on $1.25 million worth of a stock portfolio with beta = 1.5. The option delta is .8. It wishes to hedge out its resultant exposure to a market advance...
-
Write a paper about how diet relates to breast cancer in women study design to use: case control study purpose & rationale the purpose of this final project is to utilize the methods and...
-
For the object described by Figure P2.24, estimate the average velocity (a) Over the interval from t - 2.0 s to t - 4.0 s and (b) Over the interval from t - 1.0 s to t - 5.0 s. Figure P2.24 -x (m)...
-
Consider the positiontime graph shown in Figure P2.24. Make a careful graphical estimate of the velocity as a function of time by measuring the slopes of tangent lines. What is an approximate value...
-
Match each of the following examples of motion to one of the positiontime graphs in Figure P2.22. (a) A person at the beginning of a race, starting from rest (b) A runner near the end of a race, just...
-
Fill in the blanks to formulate the induction hypothesis of the proof of Claim 4.5.3: [Select] kN [Select] MEN such that if m
-
Find the 4th term of the sequences= =(n-2) (3)"-1 Blank 1
-
In each of Problems 1-10, find the eigenvalues and eigenfunctions of the regular or periodic Sturm- Liouville problem, and list a complete set of eigenfunctions. 1. y" + y = 0; y(0) = y' (L) = 0 2....

Study smarter with the SolutionInn App


