Consider the production of carbon dioxide and methane from carbon monoxide and hydrogen. Strategy Use Hesss law
Question:
Consider the production of carbon dioxide and methane from carbon monoxide and hydrogen.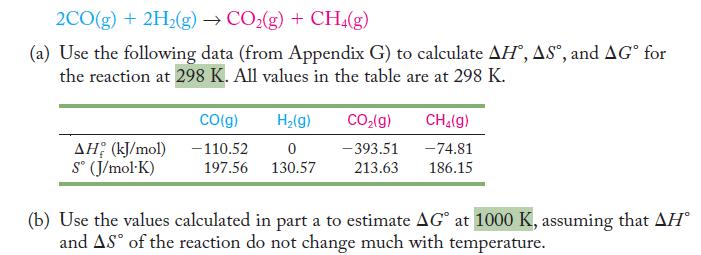
Strategy
Use Hess’s law to determine ΔH ° and ΔS °; then calculate ΔG °.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
At 1000 K the reaction is no longer spontaneous at standardstate conditions The result ...View the full answer

Answered By

Sufiyan Ahmed Tariq
I am a Chartered Accountant and an Associate Public & Finance Accountant. I also hold a bachelors of Commerce degree. I have over 8 years of experience in accounting, finance and auditing. Through out my career, I have worked with many leading multinational organisation.
I have helped a number of students in studies by teaching them key concepts of subjects like accounting, finance, corporate law and auditing. I help students understanding the complex situation by providing them daily life examples.
I can help you in the following subject / areas:
a) Accounting;
b) Finance;
c) Commerce;
d) Auditing; and
e) Corporate Law.
4.90+
7+ Reviews
17+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
The actual concentration of carbon dioxide in exhaled air is about 0.04, or 100 times the ambient concentration. Find the value of S that gives this as the equilibrium. The above problem investigates...
-
The kinetic data in the following table were obtained for the reaction of carbon dioxide and water to produce bicarbonate and hydrogen ion catalyzed by carbonic anhydrase: CO2 + H2O HCO-3 + H+ [H....
-
elow is selected financial information for SunRise Company. Selected Balance Sheet Data - As of Dec. 31, 2018 Dec. 31, 2017 Cash and short-term investments $ 958,245 $ 745,800 Accounts Receivable...
-
Treatment of an ?-amino acid with DCC yields a 2, 5-diketopiperazine. Propose a mechanism. H3N DCC H- N-H 4-N An a-amino acid A 2,5-diketopiperazine R.
-
A cat walks in a straight line, which we shall call the x-axis with the positive direction to the right. As an observant physicist, you make measurements of this cat's motion and construct a graph of...
-
The distribution of the ages of the winners of the Tour de France from 1903 to 2016 is approximately bell-shaped. The mean age is 27.9 years, with a standard deviation of 3.3 years. Use the...
-
Santos Company currently manufactures one of its crucial parts at a cost of $3.40 per unit. This cost is based on a normal production rate of 50,000 units per year. Variable costs are $1.50 per unit,...
-
9. Consider an ideal diesel cycle with compression ratio 20 and cut-off ratio 4. The maximum volume is5L. The air is at a temperature of 18 Cand pressure of 110kPa before the iso entropic...
-
Calculate the Gibbs free-energy change for the reaction of nitrogen monoxide and bromine to form nitrosyl bromide at 298 K under two sets of conditions. (a) The partial pressure of each gas is 1.0...
-
Calculate G and determine whether the following reaction will take place spontaneously under standard-state conditions at 298 K. Strategy Use the standard Gibbs free energies of formation from...
-
Can supply chain partners address the issues that are related to the existence of different standards (that also change over time)?
-
Describe the component selectors for one-dimensional arrays at the logical level.
-
The queue is implemented as a class containing an array of items, a data member indicating the index of the last item put on the queue (rear), a data member indicating the index of the location...
-
The following program has three separate errors, each of which would cause an infinite loop. As a member of the inspection team, you could save the programmer a lot of testing time by finding the...
-
1. In what cases, if any, is the bubble sort O(N)? 2. In what cases, if any, is the selection sort O(log N)? 3. In what cases, if any, is quick sort O(N )?
-
Differentiate between unit testing and integration testing.
-
Explain the different business valuation methods. Which do you think is superior and why?
-
(a) Water flows through the nozzle of a garden hose. Find an expression for m in terms of line pressure P 1 , ambient pressure P 2 , inside hose diameter D 1 , and nozzle outlet diameter D 2 . Assume...
-
Compute points on the velocity profile from the pipe wall to the centerline of a 3/4-in Type K copper tube if the volume flow rate of water at 60F is 0.50 gal/min. Use increments of 0.05 in and...
-
Compute points on the velocity profile from the tube wall to the centerline of a plastic pipe, 125 mm OD 7.4 mm wall, if the volume flow rate of gasoline (sg = 0.68) at 25C is 3.0 L/min. Use...
-
A simple heat exchanger is made by welding one-half of a 1¾-in drawn steel tube to a flat plate as shown in Fig. 9.30. Water at 40°F flows in the enclosed space and cools the plate....
-
Given the information from the table below, appraise Starbuck's cash flow from assets, profitability and long-term solvency ratios over the last three years. Cash flow from assets 2020 2021 2022 $76...
-
CarMax is a publicly-traded car dealership. Most car dealerships use debt as part of their capital structure... the industry average debt-to-value ratio is 26%. However, CarMax has no debt in their...
-
After reviewing your expenses and income, provide a one half pg essay explaining how you can improve your financial situation. What do you need to consider to decrease your expenses and/or increase...

Study smarter with the SolutionInn App


