Draw an energy-level diagram based on each of the following thermochemical equations. Label each level with the
Question:
Draw an energy-level diagram based on each of the following thermochemical equations. Label each level with the amounts of substances present, and use an arrow between levels for the given enthalpy change.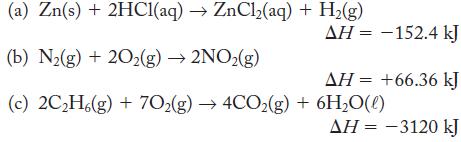
Transcribed Image Text:
(a) Zn(s) + 2HC1(aq) → ZnCl₂(aq) + H₂(g) (b) N₂(g) + 20₂(g) →2NO₂(g) (c) 2C₂H6(g) + 702(g) →4CO₂(g) + 6H₂O(l) AH = -152.4 kJ ΔΗ = +66.36 kJ AH = -3120 kJ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
While I cannot directly draw an energylevel diagram for you I can certainly explain how you could construct one for each of these thermochemical equat...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Draw an energy-level diagram based on each of the following thermochemical equations. Label each level with the amounts of substances present, and use an arrow between levels for the given enthalpy...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Hyten Corporation On June 5, 1998, a meeting was held at Hyten Corporation, between Bill Knapp, Director of Marketing/Sales, and John Rich, director of engineering. The purpose of the meeting was to...
-
The graph of f is given. (a) Why is f one-to-one? (b) What are the domain and range of f 1 ? (c) What is the value of f 1 (2)? (d) Estimate the value of f -1 (0). 1
-
Nitrocellulose fibers with an initial total water content of 40 wt% (dry basis) are dried in trays in a tunnel dryer operating at 1 atm. If the fibers are brought to equilibrium with air at 25?C and...
-
Vaughn Manufacturing purchased a one-year insurance policy in January 2017 for $36720. The insurance policy is in effect from March 2017 through February 2018. If the company neglects to make the...
-
Should additional laminate be purchased. If so, how much? LO.1
-
Hot water at 50C is routed from one building in which it is generated to an adjoining building in which it is used for space heating. Transfer between the buildings occurs in a steel pipe (k = 60 W...
-
Pine corporation has the following accounts at December 31 the common stock 10 par 5000 shares issued 50,000 paid in capital in excess of par common stock 30,000 retained earnings 45,000 and treasury...
-
Tuckered Outfitters plans to market a custom brand of packaged trail mix. The ingredients for the trail mix will include Raisins, Grain, Chocolate Chips, Peanuts, and Almonds costing, respectively,...
-
Using the following thermochemical equations 7 CH6(g) + O(g)2CO(g) + 3HO(l) 2CH(g) + 50(g) 4CO2(g) +2HO(l) H(g) + 0(g) HO(l) calculate AH for AH = -1560 kJ CH(g) + 2H(g) CH6(g) = -2599 kJ = -286...
-
A 0.470-g sample of magnesium reacts with 200 g dilute HCl in a coffee-cup calorimeter to form MgCl 2 (aq) and H 2 (g). Th e temperature increases by 10.9 C as the magnesium reacts. Assume that the...
-
In Problems 942, find each limit algebraically. lim 5x + 4 x-1
-
Use the following information for questions 1 and 2. Caterpillar Financial Services Corp. (a subsidiary of Caterpillar) and Sterling Construction sign a lease agreement dated January 1, 2020, that...
-
Identifying Binomial Distributions. Determine whether the given procedure results in a binomial distribution or a distribution that can be treated as binomial (by applying the 5% guideline for...
-
Case 6: TOMS Shoes in 2016: An Ongoing Dedication to Social Responsibility, by Margaret A. Peteraf, Sean Zhand, and Meghan L. Cooney (page C-57) Read the case and then respond to the case questions...
-
Quatro Co. issues bonds dated January 1, 2019, with a par value of $740,000. The bonds' annual contract rate is 13%, and interest is paid semiannually on June 30 and December 31. The bonds mature in...
-
Wildcat Mining wants to know the appropriate discount rate to use in their capital budgeting decision making process. Based on the following data, what is the weighted average cost of capital the CFO...
-
Figure 23.6 shows the log corresponding to a particular schedule at the point of a system crash for the four transactions T1, T2, T3, and T4 of Figure 19.4. Suppose that we use the immediate update...
-
How can a promoter avoid personal liability for pre-incorporation contracts?
-
Express a in terms of a + and a for (a) Li 2 CO 3 (b) CaCl 2 (c) Na 3 PO 4 (d) K4 F e(CN) 6 . Assume complete dissociation.
-
Calculate G o solvation in an aqueous solution for Rb + (aq) using the Born model. The radius of the Rb + ion is 161 pm.
-
Calculate the ionic strength in a solution that is 0.0750 m in K 2 SO 4 , 0.0085 m in Na 3 PO 4 , and 0.0150 m in MgCl 2 .
-
Mass LLp developed software that helps farmers to plow their fiels in a mannyue sthat precvents erosion and maimizes the effoctiveness of irrigation. Suny dale paid a licesnsing fee of $23000 for a...
-
Average Rate of Return The following data are accumulated by Lone Peak Inc. in evaluating two competing capital investment proposals: 3D Printer Truck Amount of investment $40,000 $50,000 Useful life...
-
4. (10 points) Valuation using Income Approach An appraiser appraises a food court and lounge and provides the following assessment: o O The building consists of 2 floors with the following (6)...

Study smarter with the SolutionInn App


